Kinetic Theory of Gases: How Molecular Motion Explains Pressure and Temperature
Right now, the air molecules around you are moving at about 500 meters per second. That’s faster than most bullets. Yet you don’t feel a thing. No impact. No wind. Nothing.
Why? Because these molecules are unimaginably tiny and they’re crashing into each other billions of times per second, travelling only about 70 nanometers before the next collision. The smell of perfume takes minutes to cross a room — even though each molecule in that perfume is moving at supersonic speed.
This strange reality is explained by one of the most powerful ideas in physics: the kinetic theory of gases. It connects what you can measure — pressure, temperature, volume — to what you can’t see: trillions of tiny molecules bouncing around in constant, random motion.
Let’s break it down from scratch.
What Is Kinetic Theory, Really?
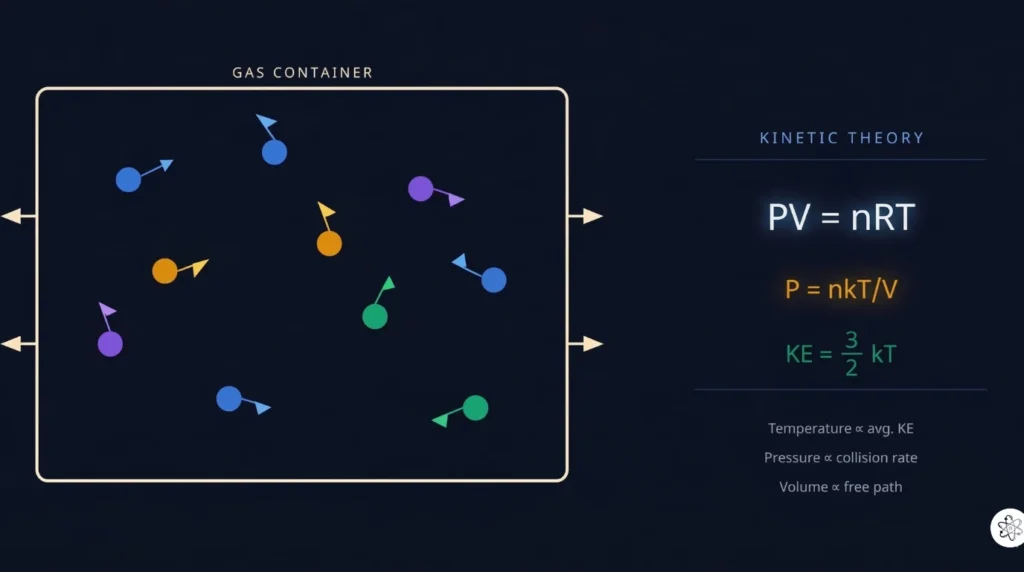
Kinetic theory is a simple but powerful idea: a gas is just a huge collection of tiny particles (atoms or molecules) flying around randomly at high speeds, constantly colliding with each other and with the walls of whatever container they’re in.
That’s it. No mysterious forces. No hidden energy fields. Just particles moving and bouncing.
From this one idea — particles in motion — we can explain why tires inflate, why pressure cookers work, why hot air balloons rise, and what temperature actually means at the molecular level.
The word “kinetic” itself comes from the Greek word kinesis, meaning motion. Kinetic theory is literally the “theory of motion” applied to gases.
The Five Assumptions Behind the Model
Before we derive anything, we need to set up the rules. Kinetic theory works with an idealized version of a gas called an ideal gas. Here are the five assumptions:
First, a gas is made up of a very large number of tiny molecules in constant, random motion. They move in straight lines until they hit something.
Second, the molecules are so small compared to the distances between them that we treat them as points with no volume. Think of it this way: if each molecule were a tennis ball, the nearest neighbour would be about 3 kilometres away.
Third, collisions are perfectly elastic. This means no kinetic energy is lost when molecules bounce off each other or off the walls. The total energy before the collision equals the total energy after.
Fourth, molecules don’t attract or repel each other. Between collisions, they travel in straight lines, completely ignoring their neighbours.
Fifth, the average kinetic energy of the molecules depends only on temperature. Higher temperature means faster molecules. Lower temperature means slower molecules.
These are simplifications, of course. Real gas molecules do have volume, and they do attract each other slightly. But at normal temperatures and pressures — the conditions you encounter in everyday life — these assumptions work remarkably well.
Where Does Pressure Come From? (The Intuition)
Imagine standing in a hailstorm. Each hailstone that hits your umbrella pushes it slightly. One hailstone barely matters. But thousands hitting every second create a steady, measurable force. That force spread across the area of your umbrella is pressure.
Gas pressure works exactly the same way.
Trillions of gas molecules slam into the walls of their container every second. Each molecule is tiny, and each individual impact delivers a minuscule push. But added together, those impacts create a constant, smooth, measurable pressure.
This is why a balloon stays inflated. Air molecules inside are continuously bombarding the inner surface of the balloon, pushing outward. The rubber pushes back. Equilibrium.
If you heat the balloon, the molecules move faster. They hit the walls harder and more often. Pressure goes up. If the rubber can’t stretch enough, the balloon pops. That’s why a balloon left in a hot car explodes — the molecular bombardment becomes too intense for the material to contain.
Deriving Pressure from Molecular Motion
Now let’s put math to the intuition. Consider a single molecule of mass m bouncing back and forth inside a cubic box of side length L.
When the molecule hits a wall and bounces back elastically, it reverses its velocity in that direction. The change in momentum from one collision is 2mv (where v is the speed in that direction). The molecule travels a distance of 2L between consecutive hits on the same wall, so the time between impacts is 2L/v.
Force from one molecule on one wall equals the rate of change of momentum: (2mv) / (2L/v) = mv²/L.
Now scale this up. A real gas has N molecules, all moving at different speeds and in random directions. On average, one-third of their motion is directed at any one pair of walls (because there are three independent directions: x, y, and z). So the total force on one wall, and therefore the pressure on that wall (force divided by the area L²), works out to:
PV = ⅓ Nm⟨v²⟩
where ⟨v²⟩ is the mean of the squared speeds of all the molecules.
This single equation is the central result of kinetic theory. It says: pressure times volume equals one-third of the total molecular kinetic energy (times 2). Pressure isn’t some separate concept from motion — it is motion, measured from the outside.
What Temperature Actually Means
Here’s where the kinetic theory delivers its most profound insight.
We already know the ideal gas law from experiments: PV = nRT, where n is the number of moles, R is the universal gas constant (8.314 J/mol·K), and T is the absolute temperature in Kelvin.
Now compare this to the kinetic theory result: PV = ⅓ Nm⟨v²⟩.
Set them equal. Do the algebra. You get:
½ m⟨v²⟩ = (3/2) k_B T
where k_B is Boltzmann’s constant (1.38 × 10⁻²³ J/K).
Read that equation carefully. The left side is the average kinetic energy of a single molecule. The right side is proportional to temperature. This means:
Temperature is not “how much heat something has.” Temperature is a direct measure of how fast the molecules are moving — their average kinetic energy.
When you heat a gas, you’re not adding some invisible substance called “heat” to it. You’re making the molecules move faster. When you cool a gas, the molecules slow down. At absolute zero (0 K, or −273.15°C), the molecules would have zero translational kinetic energy — they would stop completely. (Quantum mechanics says there’s a tiny bit of residual zero-point energy even at absolute zero, but classically, the motion stops.)
This is why temperature can never go below absolute zero. You can’t have less than zero motion.
How Fast Are Gas Molecules? The RMS Speed
From the temperature-energy equation, we can solve for speed. The result is the root mean square (RMS) speed — a kind of average speed that properly accounts for the fact that we’re dealing with squared velocities:
v_rms = √(3k_BT / m) = √(3RT / M)
where M is the molar mass in kg/mol.
Let’s calculate this for nitrogen (N₂), which makes up 78% of the air you breathe. Nitrogen has a molar mass of 0.028 kg/mol. At room temperature (293 K):
v_rms = √(3 × 8.314 × 293 / 0.028) v_rms ≈ 511 m/s
That’s about 1,840 km/h — faster than the speed of sound (343 m/s). The molecules in the air around you right now are moving at supersonic speeds.
Now try hydrogen (H₂), the lightest gas, with M = 0.002 kg/mol at the same temperature:
v_rms = √(3 × 8.314 × 293 / 0.002) v_rms ≈ 1,920 m/s
Hydrogen molecules are nearly four times faster than nitrogen molecules at the same temperature. This makes sense: lighter molecules need to move faster to have the same kinetic energy as heavier ones. It’s the same reason a ping pong ball moves much faster than a bowling ball when both have the same kinetic energy.
This also explains why hydrogen and helium slowly escape Earth’s atmosphere. They move so fast that some reach escape velocity and drift into space. Over billions of years, Earth has lost almost all of its original hydrogen and helium to space. The Moon, with its much weaker gravity, lost everything — which is why it has no atmosphere at all.
Not All Molecules Move at the Same Speed
Here’s something crucial that many students miss: the speeds given above are averages. In reality, the molecules in a gas move at a huge range of different speeds at any given moment.
Some molecules are nearly stationary. Others are moving at several times the average speed. Most are clustered near the average. This distribution of speeds is described by the Maxwell-Boltzmann distribution, one of the most important results in statistical physics.
The distribution has a distinctive shape: it rises quickly from zero, peaks at a “most probable speed” (which is slightly less than the RMS speed), and then gradually tails off toward very high speeds. The tail never quite reaches zero — there are always a few molecules moving extremely fast.
Here’s why this matters: chemical reactions require molecules to collide with at least a certain minimum energy (called the activation energy). At room temperature, the average molecule might not have enough energy to react. But the fast-moving molecules in the tail of the distribution do. Raise the temperature, and the whole distribution shifts to the right — more molecules enter the high-speed tail, and the reaction speeds up dramatically.
This is why food spoils faster in summer than in winter. The higher temperature doesn’t just make molecules “a little” faster. It exponentially increases the fraction of molecules with enough energy to drive the chemical reactions of decay.
Internal Energy of an Ideal Gas
The total kinetic energy of all the molecules in a gas is called its internal energy. For an ideal monatomic gas (like helium or argon, where the molecules are single atoms), internal energy is purely translational kinetic energy:
U = (3/2) Nk_BT = (3/2) nRT
This tells us something remarkable: the internal energy of an ideal gas depends only on temperature. Not on pressure. Not on volume. Only temperature.
Double the temperature (in Kelvin), and you exactly double the internal energy. This simple relationship is why ideal gases are so useful in physics — they give clean, predictable results that help us understand the fundamentals before we add real-world complications.
For diatomic gases like nitrogen (N₂) and oxygen (O₂), the molecules can also rotate and vibrate, which adds extra ways to store energy. These are called degrees of freedom. A monatomic gas has 3 degrees of freedom (motion in x, y, z). A diatomic gas has 5 at room temperature (3 translational + 2 rotational). Each degree of freedom contributes ½k_BT of energy per molecule, so diatomic gases have higher internal energy at the same temperature:
U = (5/2) nRT (for diatomic gases at room temperature)
The Ideal Gas Law — Derived from Scratch
The ideal gas law (PV = nRT) was discovered through experiments by Boyle, Charles, and Gay-Lussac long before anyone knew about molecules. They measured pressure, volume, and temperature and noticed patterns.
Kinetic theory provides the reason behind those patterns. Starting from nothing but Newton’s laws, elastic collisions, and the identification of temperature with kinetic energy, we derived PV = ⅓Nm⟨v²⟩ and then showed that ½m⟨v²⟩ = (3/2)k_BT.
Combining these:
PV = ⅓N · 2 · (3/2)k_BT = Nk_BT
Since Nk_B = nR (where n is moles and R is the gas constant), we get:
PV = nRT
This derivation is one of the most satisfying in all of physics. A law that was found by experiment — by measuring gas in flasks and tubes — turns out to be a mathematical consequence of tiny invisible particles bouncing off walls. Thermodynamics and mechanics are not separate subjects. They are two ways of looking at the same thing.
Visual Explanation
Real-World Examples That Make It Click
Why does a pressure cooker work? When you seal a pot and heat it, the water molecules turn to steam and move faster. They can’t escape, so they hit the walls more often and harder. Pressure inside increases well above atmospheric pressure. Higher pressure raises the boiling point of water above 100°C, so food cooks faster.
Why does a car tire pressure increase after driving? Friction between the tires and road heats the air inside the tire. Hotter air means faster molecules, which means more forceful collisions with the tire walls. The pressure goes up — sometimes by 5–10 psi on a long highway drive.
Why does a helium balloon deflate faster than an air balloon? Helium atoms are much smaller and lighter than nitrogen and oxygen molecules. They move faster (higher RMS speed at the same temperature) and slip through the tiny pores in the balloon rubber more easily. A helium balloon left overnight is noticeably smaller by morning.
Why is the sky mostly nitrogen and oxygen but almost no hydrogen? Hydrogen is the lightest gas, so it moves the fastest. At the top of the atmosphere, some hydrogen molecules reach escape velocity (about 11.2 km/s for Earth). Over billions of years, hydrogen has escaped into space. Heavier gases like nitrogen (28 g/mol) and oxygen (32 g/mol) move too slowly to escape.
Why does spray feel cold when it hits your skin? The gas in an aerosol can is compressed liquid. When it’s released, it expands rapidly. The molecules spread out and do work against intermolecular forces, losing kinetic energy. Lower kinetic energy means lower temperature. The spray feels cold because the molecules literally slowed down.
Common Mistakes Students Make
Mistake 1: “Temperature is the amount of heat in an object.” This is wrong. Temperature is the average kinetic energy of the molecules — how fast they move. Heat is the transfer of energy from a hotter object to a cooler one. An object can have a high temperature but very little total thermal energy (like a tiny spark), and vice versa (like a lukewarm swimming pool, which has enormous total energy despite its modest temperature).
Mistake 2: “Heavier molecules move faster because they have more energy.” The opposite is true. At the same temperature, all gas molecules have the same average kinetic energy (½m⟨v²⟩ = 3/2 k_BT). Since KE = ½mv², heavier molecules must move slower to have the same energy. Lighter molecules move faster. This is exactly what the RMS speed formula shows — speed is inversely proportional to the square root of mass.
Mistake 3: “All molecules in a gas move at the same speed.” They don’t. Speeds follow the Maxwell-Boltzmann distribution — a wide range from nearly zero to several times the average. The RMS speed is a useful average, but real gases always contain some very fast and some very slow molecules at any instant.
Mistake 4: “Pressure means molecules pushing each other outward.” Pressure comes from molecules hitting the walls of the container, not from molecules pushing on each other. In fact, one of the ideal gas assumptions is that molecules don’t exert forces on each other between collisions. The pressure is wall bombardment, not internal crowding.
Mistake 5: “If I double the Celsius temperature, pressure doubles.” No. Gas laws use absolute temperature (Kelvin), not Celsius. Doubling from 20°C to 40°C is going from 293 K to 313 K — an increase of only about 7%, not 100%. To actually double the pressure (at constant volume), you’d need to double the Kelvin temperature: from 293 K to 586 K (313°C).
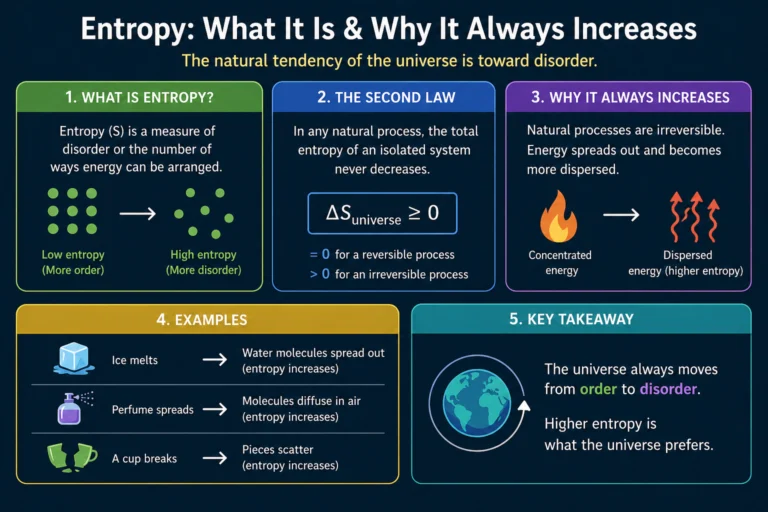
Quick Summary
- Kinetic theory explains gas behavior by treating gas as a huge collection of tiny molecules in constant, random motion.
- Pressure is caused by molecules colliding with container walls — trillions of tiny impacts per second creating a measurable force.
- Temperature is the average translational kinetic energy of the molecules. Higher temperature means faster molecules. Absolute zero means zero motion.
- The central equation PV = ⅓Nm⟨v²⟩ connects pressure and volume to molecular speed, and leads directly to the ideal gas law PV = nRT.
- RMS speed (v_rms = √(3RT/M)) shows that lighter molecules move faster than heavier ones at the same temperature.
- Molecular speeds aren’t uniform — they follow the Maxwell-Boltzmann distribution, with a long high-speed tail that explains why reactions speed up dramatically with temperature.
- The internal energy of an ideal gas depends only on temperature, not on pressure or volume.
- These concepts explain real-world phenomena from tire pressure to why Earth’s atmosphere lost its hydrogen.
Frequently Asked Questions
What is kinetic theory of gases in simple words?
Kinetic theory says that gas is made of tiny particles (molecules) that are constantly moving in random directions at very high speeds. The things we can measure — pressure, temperature, volume — are all results of this invisible molecular motion. Pressure comes from molecules hitting walls. Temperature measures how fast the molecules are moving on average.
Why do gas molecules move so fast if we can’t feel them?
Each molecule is incredibly tiny — about 10⁻¹⁰ meters across. Even though they move at hundreds of meters per second, their mass is so small that each individual impact is negligible. You feel the collective effect as air pressure, but not the individual hits. It’s like rain: one raindrop barely registers, but a storm is impossible to ignore.
What is the difference between temperature and heat?
Temperature is the average kinetic energy of molecules — a property of the substance itself. Heat is the transfer of energy from a hotter object to a cooler one — it’s a process, not a property. A spark has high temperature but almost no heat to transfer. A bathtub of warm water has moderate temperature but a lot of total thermal energy.
Why does temperature use Kelvin in gas laws?
Celsius and Fahrenheit have arbitrary zero points. Kelvin starts at absolute zero — the point where molecular motion stops. Gas laws describe proportional relationships (double the temperature, double the pressure), and those proportions only work correctly with a scale where zero actually means zero. Using Celsius would give nonsensical results, like predicting that 0°C means zero pressure.
Can molecules really reach escape velocity and leave Earth?
Yes. The Maxwell-Boltzmann distribution means a small fraction of molecules are always moving much faster than the average. For light gases like hydrogen and helium, this fraction includes molecules moving faster than 11.2 km/s — Earth’s escape velocity. Over geological time, enough molecules escape to strip these gases from the atmosphere entirely. This is called atmospheric escape, or Jeans escape.
What happens at absolute zero?
Classically, all molecular motion stops at 0 K (−273.15°C). In reality, quantum mechanics says particles retain a small amount of residual energy called zero-point energy even at absolute zero, so they never truly stop. But absolute zero is the lowest possible temperature — you can’t remove more kinetic energy than a molecule has.
Why doesn’t the ideal gas law work at very high pressures or low temperatures?
At high pressures, molecules are squeezed so close together that their actual volume matters — they can no longer be treated as points. At low temperatures, intermolecular attractive forces (like van der Waals forces) become significant relative to the reduced kinetic energy. Both of these violate the ideal gas assumptions. Real gases under these conditions are better described by the van der Waals equation, which includes corrections for molecular volume and attractive forces.
Articles from same category
- kinetic theory of gases
- First Law of Thermodynamics
- What is Energy
- Second Law of Thermodynamics
Frequently Asked Questions
Get physics insights delivered weekly
Join students and educators receiving expert explanations, study tips, and platform updates every Thursday.
Join others. No spam.
allphysicsfundamentals
Making physics accessible, interactive, and genuinely understandable for students at every level.
Learn
- All Articles
- Classical Mechanics
- Thermodynamics
- Waves & Optics
- Electromagnetism
- Modern Physics