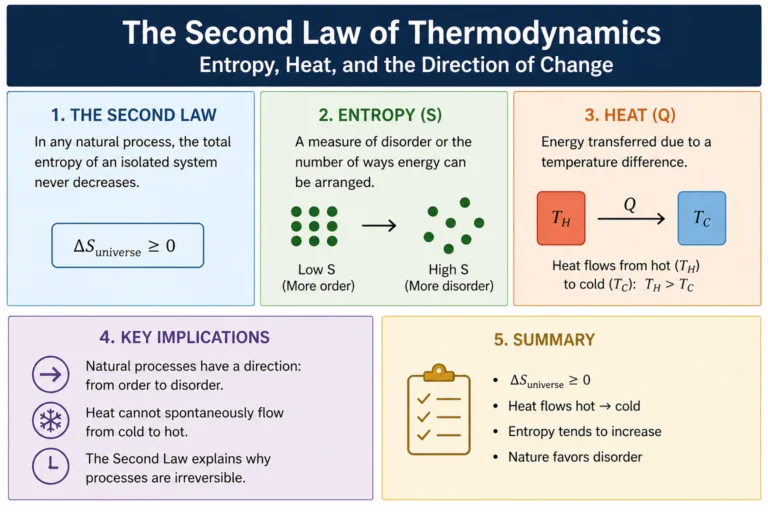
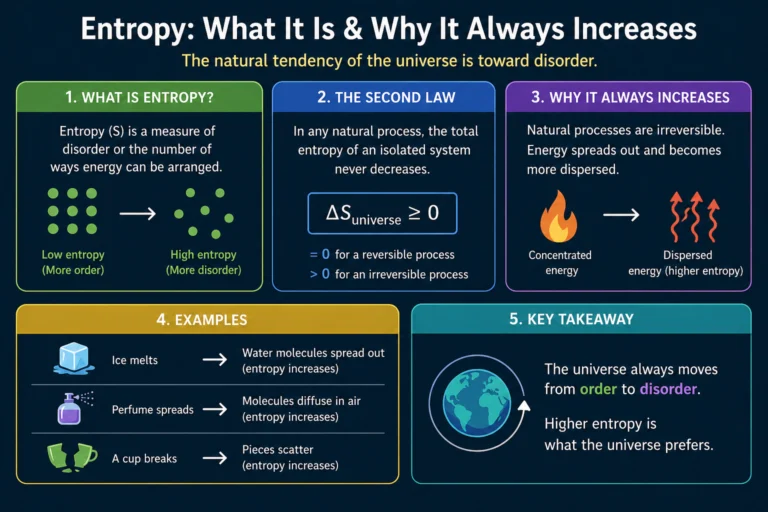
Master the Ideal Gas Law: The Easy Guide to PV = nRT
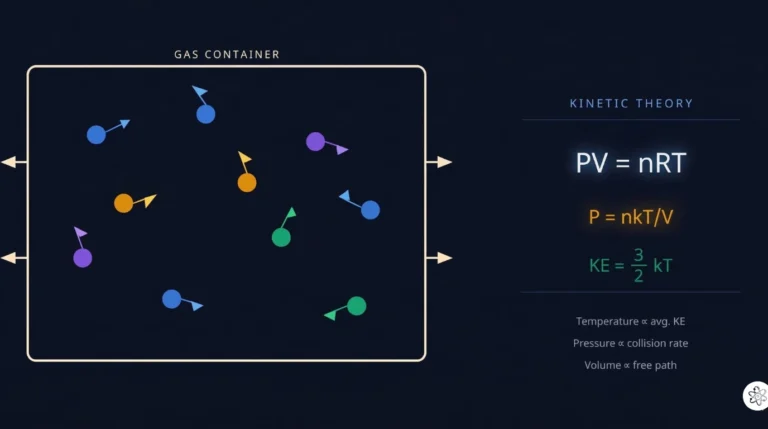
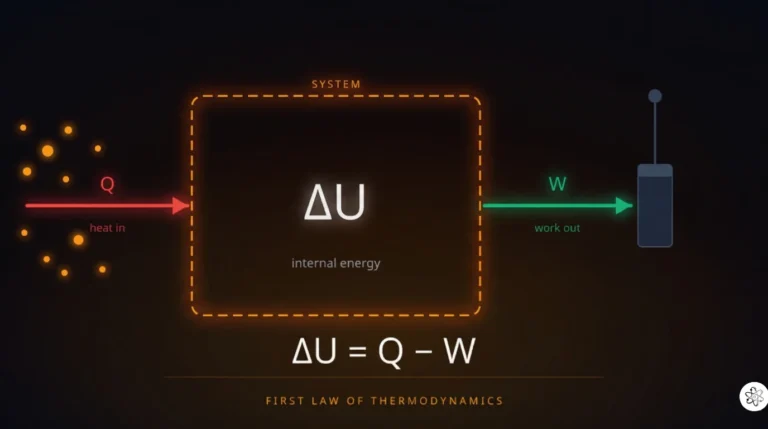
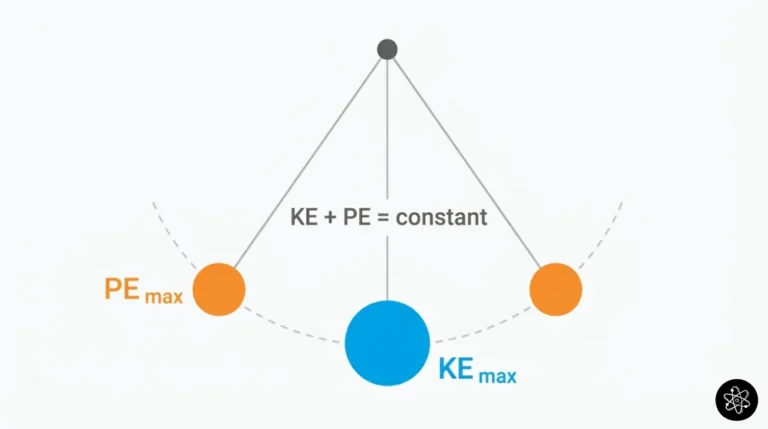
Wondering why balloons expand when heated or how tire pressure works? Unlock the secrets of gas behavior with our easy-to-understand guide to the Ideal Gas Law ($PV = nRT$). We break down the formula, show you real-world examples, and help you ace your chemistry quiz!