The First Law of Thermodynamics: Why Energy Always Balances
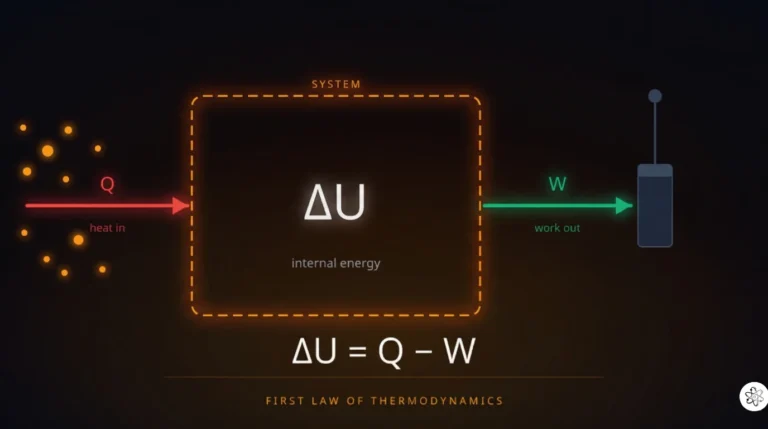
Every machine you’ve ever used — your car engine, your refrigerator, even your own body — obeys one rule above all others. Energy goes in, energy comes out, and the books always balance. That’s the first law of thermodynamics, and it’s the reason perpetual motion machines don’t exist, why your car needs fuel, and why you get hungry after exercise.
It sounds simple. It is simple — at its core. But the way it plays out across different physical processes is where things get interesting.
The Equation: ΔU = Q − W(The First Law of Thermodynamics)
The first law of thermodynamics says: the change in a system’s internal energy equals the heat added to the system minus the work the system does on its surroundings.
ΔU = Q − W
Let’s break that down piece by piece.
ΔU is the change in internal energy — the total microscopic energy stored inside your system. Think of it as the energy “balance” in the system’s bank account.
Q is heat — energy flowing into or out of the system because of a temperature difference. Q is positive when heat flows in, negative when it flows out.
W is work — energy the system spends pushing against its surroundings (like a gas expanding against a piston). W is positive when the system does work on the outside, negative when work is done on the system.
That’s it. Every joule that enters the system as heat either stays inside (increasing internal energy) or leaves as work. Nothing appears from thin air. Nothing vanishes. The first law is energy conservation applied to systems where heat and work are the currencies of energy transfer.
What Exactly Is Internal Energy?
Internal energy is the sum of everything happening at the molecular level inside your system. Molecules zipping around (translational kinetic energy), molecules spinning (rotational energy), atoms within molecules vibrating back and forth (vibrational energy), and the potential energy from forces between neighboring molecules.
You can’t measure internal energy directly — you don’t have a thermometer that reads “total molecular kinetic energy.” But you can measure changes in it through temperature, pressure, and volume changes. And for an ideal gas (the simplified model you’ll use in most physics courses), internal energy depends only on temperature:
U = nCvT
Where n is the number of moles, Cv is the molar heat capacity at constant volume, and T is the temperature in kelvin. Raise the temperature of an ideal gas and you’ve raised its internal energy. Lower the temperature and you’ve lowered it. No exceptions.
This also means that for an ideal gas, if the temperature doesn’t change, the internal energy doesn’t change — regardless of what else is going on. That fact is surprisingly powerful when you start analyzing thermodynamic processes.
Heat and Work Are Not the Same Thing
This is one of those conceptual traps that catches a lot of students. Heat and work both transfer energy, but they do it in fundamentally different ways.
Heat is energy transfer driven by a temperature difference. Put a hot object next to a cold one and energy flows from hot to cold automatically. You don’t have to do anything — the temperature gradient does the work (figuratively speaking). Heat flows through conduction, convection, or radiation.
Work is energy transfer through a macroscopic force acting over a distance. A gas pushing a piston outward does work on the piston. You compressing a gas with your hand does work on the gas. It’s mechanical, it’s directed, and it involves something actually moving.
Here’s the crucial distinction that trips people up: heat and work are processes, not things a system contains. You can’t say “this gas contains 500 joules of heat.” That’s meaningless. You can say “500 joules of heat were transferred to the gas.” Internal energy is what the system has. Heat and work are how it gets or loses that energy.
It’s like your bank account. You have a balance (internal energy). You can deposit money (heat in) or withdraw it (work out). But the money in your wallet during the transaction isn’t “in the account” — it’s in transit. Heat and work are the deposits and withdrawals. Internal energy is the balance.
The Four Key Thermodynamic Processes
The first law applies universally, but it simplifies in beautiful ways depending on what you hold constant. There are four special cases that show up everywhere in physics and engineering.
Isothermal — temperature stays constant
If the temperature doesn’t change, then for an ideal gas, ΔU = 0. The first law becomes Q = W. Every bit of heat you add goes straight into work, and vice versa. This happens when a gas expands or compresses slowly enough to stay in thermal equilibrium with its surroundings. Isothermal expansion is a key step in the idealized Carnot engine cycle.
Adiabatic — no heat transfer
When Q = 0, the first law simplifies to ΔU = −W. The system changes its internal energy purely through work. Compress a gas adiabatically and it heats up — that’s how diesel engines work. The air gets compressed so fast and so much that it reaches temperatures high enough to ignite fuel without a spark plug. Let a gas expand adiabatically and it cools — that’s the principle behind the cooling you feel when air rushes out of a pressurized tire.
Isochoric — volume stays constant
No volume change means no work (since W = PΔV and ΔV = 0). So ΔU = Q — all heat goes into changing the internal energy. Heat a sealed rigid container and the gas inside just gets hotter. This is the simplest case and shows up in bomb calorimeters and constant-volume gas thermometers.
Isobaric — pressure stays constant
At constant pressure, both Q and W are non-zero, so you use the full first law equation. Heating water in an open pot is roughly isobaric — the water absorbs heat, does a little work expanding against atmospheric pressure, and increases its internal energy. Most chemical reactions and biological processes happen at roughly constant atmospheric pressure, so isobaric analysis is extremely common in chemistry and biology.
Visual Explanation of the First Law of Thermodynamics
Process type
Heat (Q)
+200 J
Work (W)
+80 J
ΔU (internal energy)
+120 J
ΔU = 200 − 80 = +120 J
Gas heats up — internal energy increases
Why Perpetual Motion Machines Can’t Exist
The first law kills one category of perpetual motion machine right away: the kind that claims to produce energy from nothing. If ΔU = Q − W and no heat flows in (Q = 0), then W = −ΔU. You can only do work by draining your internal energy. Once it’s gone, you stop. You can’t create energy out of nowhere.
People have been trying to build these machines for centuries. Every single one either secretly takes in energy from somewhere (a hidden heat source, the environment) or simply doesn’t work. The first law says this isn’t a practical limitation — it’s a fundamental one. Energy conservation isn’t something that could be broken with cleverer engineering. It’s baked into the structure of physics.
The second law of thermodynamics adds another layer — even when you have energy to spend, you can’t convert all of it to work. But the first law is the foundation: you can’t create it at all.
Heat Engines and Why 100% Efficiency Is Impossible
A heat engine takes in heat from a hot source, converts some of it to work, and dumps the rest into a cold sink. Your car engine does this: it burns fuel (hot source), pushes pistons (work), and exhausts waste heat through the radiator and tailpipe (cold sink).

Over a complete engine cycle, the system returns to its starting state, so ΔU = 0. The first law tells us Qnet = W for the cycle. The work you get out equals the net heat flowing through the system. But here’s the thing — “net heat” is Qin − Qout. Some heat always gets exhausted. The second law proves you can never avoid this — there’s a theoretical maximum efficiency set by the Carnot limit:
ηmax = 1 − Tcold/Thot
Where the temperatures are in kelvin. A car engine running between about 2500 K combustion temperature and 300 K exhaust temperature has a theoretical maximum efficiency around 88%. Real engines get nowhere near that because of friction, heat losses, and incomplete combustion — typical gasoline engines run around 20-30% efficiency. But the first law tells you why the ceiling exists, and the second law tells you where the ceiling is.
Your Body Runs on the First Law Too
Your body is a thermodynamic system. Food is your heat source — the chemical bonds in carbohydrates, fats, and proteins store internal energy. Metabolism breaks those bonds and releases that energy. Some of it does mechanical work (muscle contractions, pumping blood). The rest becomes heat that your body radiates away to maintain a constant temperature of about 37°C.
When you eat a 2000-calorie diet (that’s about 8400 kilojoules), your body converts that energy into work and heat. If you exercise heavily, more goes to work and you generate more heat (which is why you sweat). If you sit on the couch, almost all of it becomes heat. The calorie counts on food labels are direct measurements of the internal energy stored in those foods — determined by literally burning the food in a calorimeter and measuring the heat released.
And if you eat more energy than you use? The excess gets stored — mostly as fat, which is just chemical potential energy your body banks for later. The first law doesn’t care about nutrition science or diets. Calories in minus calories out equals change in stored energy. It’s ΔU = Q − W with a biological coat of paint.
A Quick History: How We Figured This Out
For a long time, people thought heat was a fluid — called “caloric” — that flowed between objects. It was a reasonable idea. Heat does seem to flow from hot things to cold things, sort of like a liquid.
The caloric theory started crumbling in the 1840s. James Prescott Joule ran a series of experiments showing that mechanical work could produce heat in exact, repeatable proportions. He used a falling weight to spin a paddle wheel in water and measured the temperature rise. The conclusion was unavoidable: heat and work are both forms of energy transfer, interconvertible with a fixed exchange rate.
Rudolf Clausius put the pieces together in 1850, giving us the first explicit statement of the first law. And the mechanical equivalent of heat — Joule’s key discovery — is why we now measure both heat and work in the same units (joules, fittingly named after him).
Frequently Asked Questions
What does the first law of thermodynamics state?
It states that the change in internal energy of a system equals the heat added to it minus the work it does: ΔU = Q − W. In plain terms, energy can’t be created or destroyed — it can only be transferred as heat or work. Every joule is accounted for.
What’s the difference between heat and work?
Heat is energy transfer caused by a temperature difference — it flows spontaneously from hot to cold. Work is energy transfer through a mechanical force acting over a distance, like a gas pushing a piston. Both are processes of energy transfer, not properties a system contains.
What is internal energy?
Internal energy is the total microscopic energy stored in a system — the kinetic energy of molecules moving, rotating, and vibrating, plus the potential energy from intermolecular forces. For an ideal gas, it depends only on temperature. You can change it by adding heat or doing work on the system.
Why can’t a perpetual motion machine work?
The first law says you can’t get energy from nothing. A machine that does work without any energy input would violate conservation of energy. Once it exhausts its internal energy, it stops — and no clever design can change that. The first law makes this a fundamental impossibility, not a practical one.
How does the first law apply to the human body?
Your body takes in chemical energy through food (Q in), does mechanical work through muscle movement (W), and radiates excess energy as heat. The calorie count on food labels measures stored internal energy. If you consume more than you use, the excess is stored as fat — it’s ΔU = Q − W applied to biology.
Articles from same category
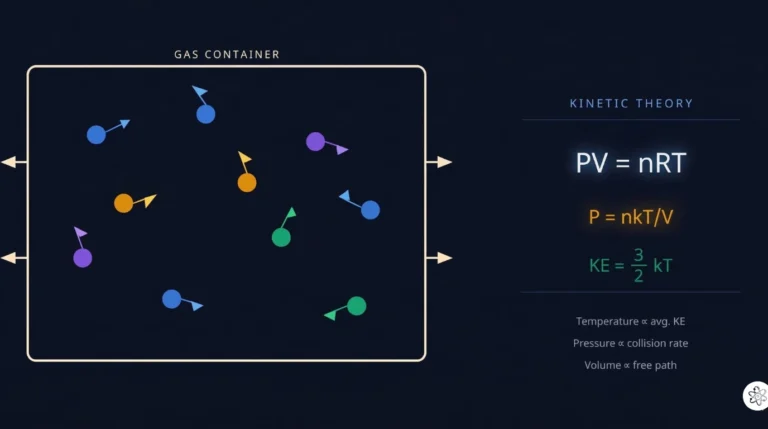
- kinetic theory of gases
- First Law of Thermodynamics
- What is Energy
- Second Law of Thermodynamics
Frequently Asked Questions
Get physics insights delivered weekly
Join students and educators receiving expert explanations, study tips, and platform updates every Thursday.
Join others. No spam.
allphysicsfundamentals
Making physics accessible, interactive, and genuinely understandable for students at every level.
Learn
- All Articles
- Classical Mechanics
- Thermodynamics
- Waves & Optics
- Electromagnetism
- Modern Physics