What Is Energy? The Physics Definition That Connects Everything
Ask a physicist to define energy, and you’ll probably get a pause. Not because they don’t know — but because energy is one of those concepts that sits so deep in the foundations of physics that pinning it down in a single sentence is harder than it sounds.
Here’s the short version: energy is a number you can calculate for any physical system, and that number never changes as long as the system is left alone. It can shift forms — from motion to heat to light to chemical bonds — but the total stays the same. That property, conservation of energy, is arguably the single most powerful idea in all of physics.
Let’s dig into what that actually means.
Energy definition in physics
In physics, energy is defined as the capacity of a system to do work. Work, in turn, is force applied over a distance. Push a box across the floor — you’ve done work, and you’ve transferred energy from your muscles to the box’s motion. The SI unit is the joule (J): one joule is the work done when a force of one newton moves something one meter.
But that “capacity to do work” definition, while technically correct, undersells what energy really is. Energy isn’t a substance. It’s not a fluid that flows between objects. It’s a property — a scalar quantity (just a number, no direction) that every physical system has, and that’s conserved in every process ever observed. It’s the universal bookkeeping system of physics.
Kinetic Energy: The Energy of Motion
Anything that moves has kinetic energy. The formula is clean and powerful:
KE = ½mv²
Two things about this equation matter a lot. First, kinetic energy depends on the square of velocity. Double your speed and your kinetic energy quadruples. A car going 100 km/h has four times the kinetic energy of the same car going 50 km/h, which is why highway crashes are so much more devastating than parking lot bumps. The energy your body has to absorb in a crash scales with speed squared, not speed.
Second, kinetic energy is always positive or zero. You can’t have negative kinetic energy because both mass and v² are always positive. An object at rest has zero KE. Everything else has a positive number.
There’s also rotational kinetic energy (½Iω² for spinning objects) and thermal kinetic energy (the random molecular jiggling that we measure as temperature). But they’re all the same idea: motion stores energy.
Potential Energy: Energy Waiting to Happen
Potential energy is stored in the arrangement of a system. Lift a ball off the ground and you’ve given it gravitational potential energy:
PE = mgh
Where m is mass, g is gravitational acceleration (9.8 m/s² on Earth), and h is height above your chosen reference point. Let go, and that stored energy converts smoothly into kinetic energy as the ball falls. At the bottom, every joule of PE has become KE. The total didn’t change — it just changed costume.
Other flavors of potential energy include elastic PE (½kx² for a compressed spring), electric PE (stored between charged particles), chemical PE (stored in molecular bonds — this is what food and fuel contain), and nuclear PE (stored in the binding forces inside atomic nuclei). They’re all “stored energy” in different configurations, ready to release when conditions change.
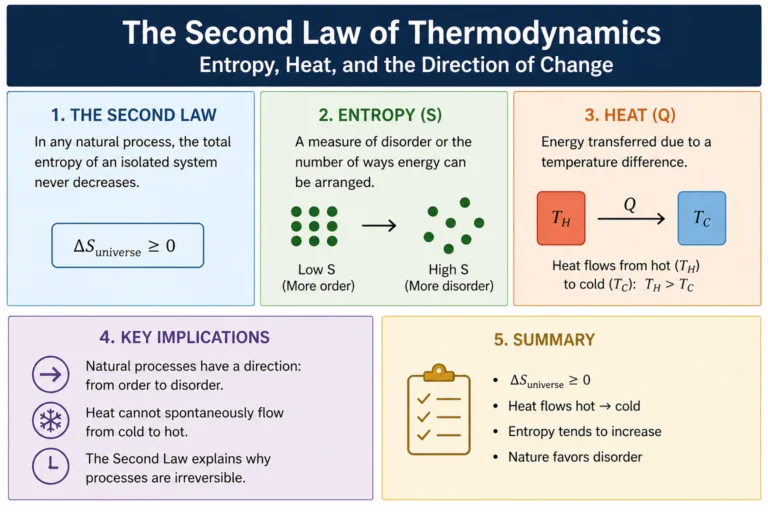
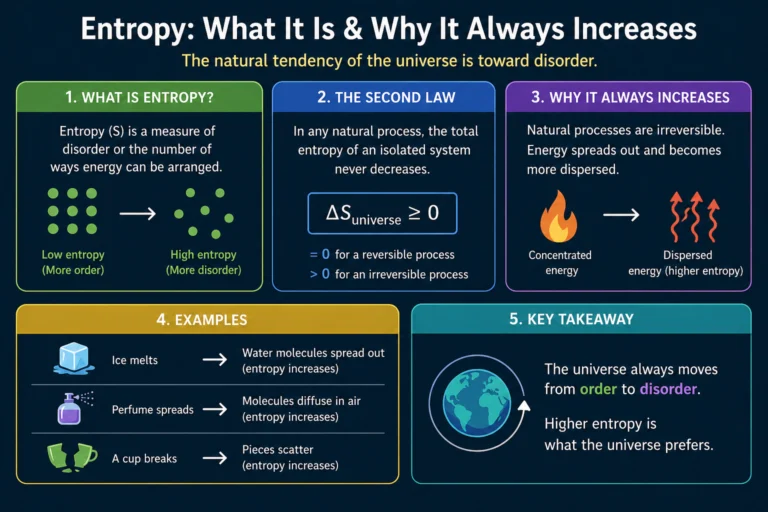
Conservation of Energy — The Law That Rules Everything
The conservation of energy states that the total energy of an isolated system never changes. It can transform — kinetic to potential, chemical to thermal, electrical to mechanical — but the total before and after is always the same. Always.
This isn’t an approximation. It isn’t a rule of thumb. It’s an exact law of nature with no known exceptions in over 200 years of testing. Every branch of physics — mechanics, thermodynamics, electromagnetism, quantum mechanics, relativity — obeys it.
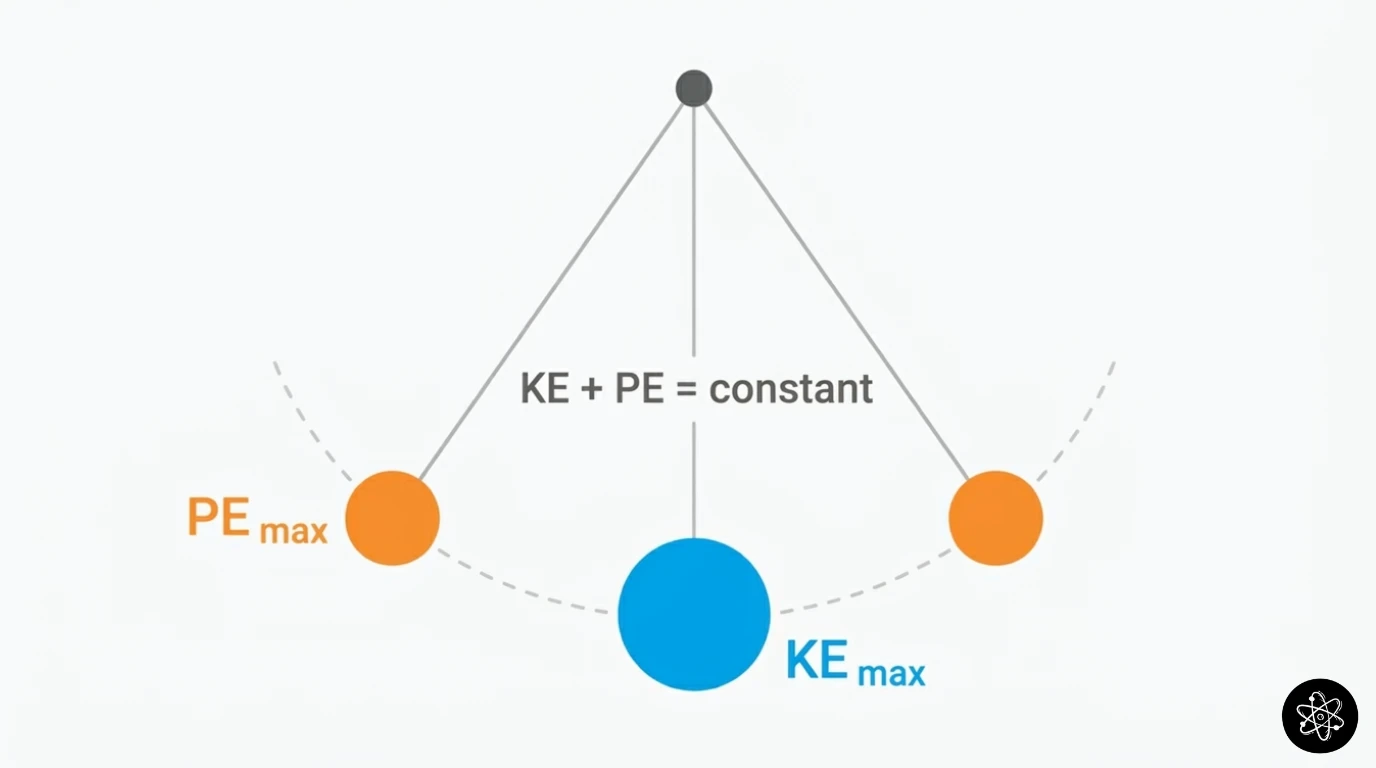
A pendulum demonstrates it beautifully. At the top of its swing: maximum PE, zero KE (momentarily stopped). At the bottom: zero PE, maximum KE (moving fastest). In between, the two forms trade back and forth, but KE + PE = constant at every point.
“But wait,” you might say, “the pendulum eventually stops. Where did the energy go?” It went into thermal energy — friction at the pivot, air resistance heating the air by a tiny amount. If you could measure the warming of the room precisely enough, you’d find the total energy — mechanical plus thermal — is still exactly conserved. Energy didn’t vanish. It just became harder to track.
Why is energy conserved? Noether’s theorem
In 1915, mathematician Emmy Noether proved something extraordinary: every symmetry in the laws of physics corresponds to a conservation law. Momentum conservation comes from the fact that physics works the same everywhere in space. Angular momentum conservation comes from the fact that physics works the same in every direction.
And energy conservation? It comes from the fact that the laws of physics don’t change over time. Run an experiment today or a billion years from now — the rules are the same. That time-translation symmetry mathematically requires energy to be conserved. It’s not an empirical accident. It’s woven into the fabric of physics itself.
The Energy Shortcut: Why Physicists Love It
Conservation of energy lets you solve problems that would be nightmarish with forces alone. Here’s a classic example.
A roller coaster starts at the top of a 30-meter hill. How fast is it going at the bottom?
Using forces (F = ma), you’d need to track the changing slope of the track, resolve forces along a curved path, and integrate. Brutal algebra.
Using energy? Two lines:
mgh = ½mv² → v = √(2gh) = √(2 × 9.8 × 30) ≈ 24.2 m/s
The mass cancels completely. Every roller coaster car, regardless of how heavy it is, hits the same speed at the bottom of the same hill (if friction is negligible). That’s the same insight Galileo discovered for falling objects — the rate of fall doesn’t depend on mass. Energy methods show why.
E = mc² — Energy and Mass Are the Same Thing
Einstein’s most famous equation says that mass itself is a form of energy:
Where c is the speed of light (3 × 10⁸ m/s). Since c² is an enormous number (9 × 10¹⁶), even a tiny amount of mass corresponds to a staggering amount of energy. One kilogram of matter, if fully converted to energy, releases about 9 × 10¹⁶ joules — roughly the energy of a 21-megaton nuclear bomb.
This isn’t theoretical — it’s practical. Nuclear power plants convert a tiny fraction of nuclear fuel mass into thermal energy. The Sun converts about 4 million tonnes of mass into energy every second through nuclear fusion. And in particle accelerators, energy routinely converts into brand-new particles — mass created from pure energy, just as E = mc² predicts.
Before Einstein, “conservation of mass” and “conservation of energy” were two separate laws. After 1905, they merged into one: conservation of mass-energy. Mass and energy are interchangeable. They’re the same thing, measured in different units.
Before Einstein, “conservation of mass” and “conservation of energy” were two separate laws. After 1905, they merged into one: conservation of mass-energy. Mass and energy are interchangeable. They’re the same thing, measured in different units.
All the Forms of Energy
Energy shows up in many disguises, but every form is either kinetic (associated with motion) or potential (associated with arrangement).
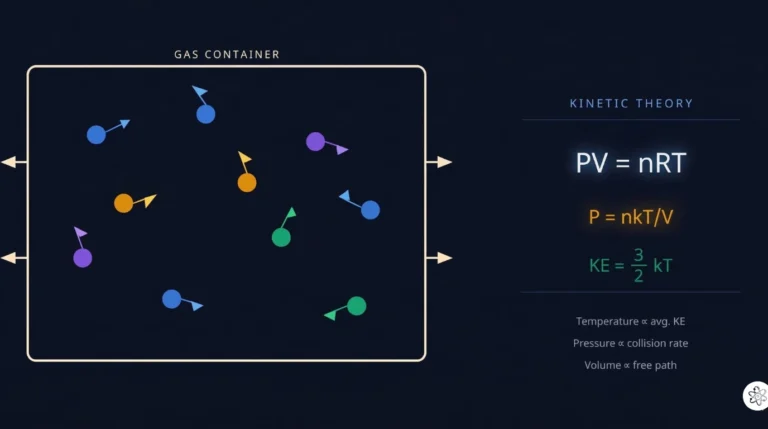
Thermal energy is the kinetic energy of molecules jiggling randomly. Temperature measures the average kinetic energy per molecule. Heat is thermal energy transferred between objects at different temperatures.
Chemical energy is potential energy stored in molecular bonds. When you eat food, your body breaks those bonds and releases energy for muscle movement and heat. When gasoline burns, the chemical bonds rearrange and release energy that moves your car.
Electrical energy is the energy of moving charges. A battery stores chemical potential energy and converts it to electrical energy — the flow of electrons through a circuit that powers your devices.
Nuclear energy is potential energy locked in the binding forces of atomic nuclei. Fission (splitting heavy atoms) and fusion (joining light atoms) both release energy because the resulting nuclei are more tightly bound — they’ve moved to a lower-energy configuration.
Electromagnetic (radiant) energy is carried by light, radio waves, X-rays, and all other electromagnetic radiation. It travels at c in vacuum and carries energy proportional to frequency: E = hf, where h is Planck’s constant. That’s why UV light gives you sunburn but radio waves don’t — UV photons carry millions of times more energy per photon.
Every physical process is just energy changing costumes. A car engine: chemical → thermal → kinetic. A solar panel: electromagnetic → electrical. Your body: chemical → kinetic + thermal. The forms change. The total doesn’t.
Visual Explanation of What Energy Is?
PE (potential)
1960 J
KE (kinetic)
0 J
Speed
0 m/s
Total energy
1960 J
Drag the sliders, then tap “Drop ball” to watch PE convert to KE
This explorer demonstrates the most fundamental idea in physics: energy is never created or destroyed — it only changes form.
A ball sits at a height you choose, loaded with gravitational potential energy (PE = mgh). Tap “Drop ball” and watch what happens:
- The purple PE bar shrinks as the ball falls — it’s losing height and therefore losing potential energy
- The orange KE bar grows at exactly the same rate — the ball is speeding up, gaining kinetic energy (KE = ½mv²)
- The green Total bar never changes — that’s conservation of energy in action. Every joule lost from PE appears as KE. Nothing is created, nothing is destroyed
Why Energy Is the Most Important Concept in Physics
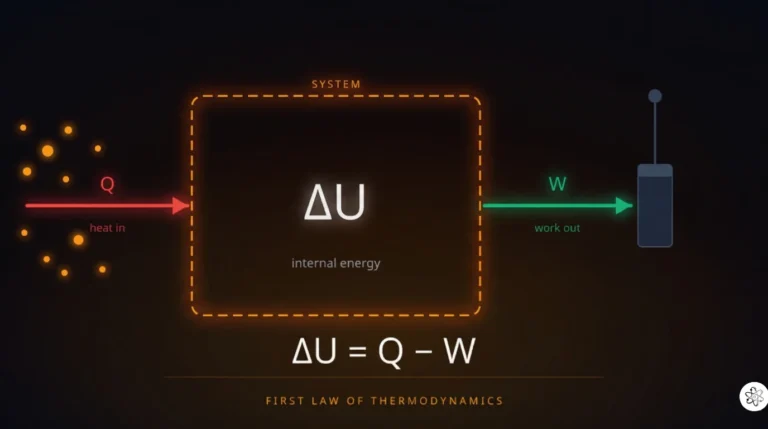
Energy shows up in every branch of physics without exception. Mechanics? KE + PE = constant. Thermodynamics? ΔU = Q − W. Electromagnetism? Power = IV. Quantum mechanics? E = hf. Relativity? E = mc². No other concept threads through every field with this kind of universality.
And energy conservation doesn’t just describe what does happen — it defines what can’t happen. Any process that would increase the total energy of an isolated system is physically impossible. Full stop. That constraint is more powerful than any specific force law because it applies to every interaction everywhere in the universe.
If you learn one thing from physics, make it this: the total energy of the universe is a constant. Everything else is just transformations.
Frequently Asked Questions
What is the definition of energy in physics?
Energy is a scalar quantity that measures a system’s capacity to do work or produce heat. It’s measured in joules (J) and exists in many forms — kinetic, potential, thermal, chemical, electrical, nuclear, electromagnetic. Its defining feature is that it’s always conserved: the total energy of an isolated system never changes.
What is the difference between kinetic and potential energy?
Kinetic energy is the energy of motion (KE = ½mv²). Potential energy is stored energy due to an object’s position or configuration (gravitational PE = mgh, elastic PE = ½kx²). They constantly convert into each other — a falling ball loses PE and gains KE. The total (KE + PE) stays constant in a frictionless system.
Can energy be created or destroyed?
No. The law of conservation of energy says energy can only change forms — never be created from nothing or destroyed into nothing. When energy seems to “disappear” (like a ball slowing down due to friction), it’s been converted to thermal energy. The total is unchanged.
What does E = mc² mean?
It means mass is a form of energy. A small amount of mass corresponds to an enormous amount of energy (because c² = 9 × 10¹⁶). This is the principle behind nuclear power and nuclear weapons — converting a tiny fraction of nuclear fuel mass into usable energy.
Why is energy conserved?
Because the laws of physics don’t change over time. Emmy Noether proved in 1915 that every symmetry in nature corresponds to a conservation law. Time-translation symmetry (physics works the same today as tomorrow) directly requires energy conservation. It’s built into the structure of the universe.
Articles from same category
- kinetic theory of gases
- First Law of Thermodynamics
- What is Energy
- Second Law of Thermodynamics
Frequently Asked Questions
Get physics insights delivered weekly
Join students and educators receiving expert explanations, study tips, and platform updates every Thursday.
Join others. No spam.
allphysicsfundamentals
Making physics accessible, interactive, and genuinely understandable for students at every level.
Learn
- All Articles
- Classical Mechanics
- Thermodynamics
- Waves & Optics
- Electromagnetism
- Modern Physics