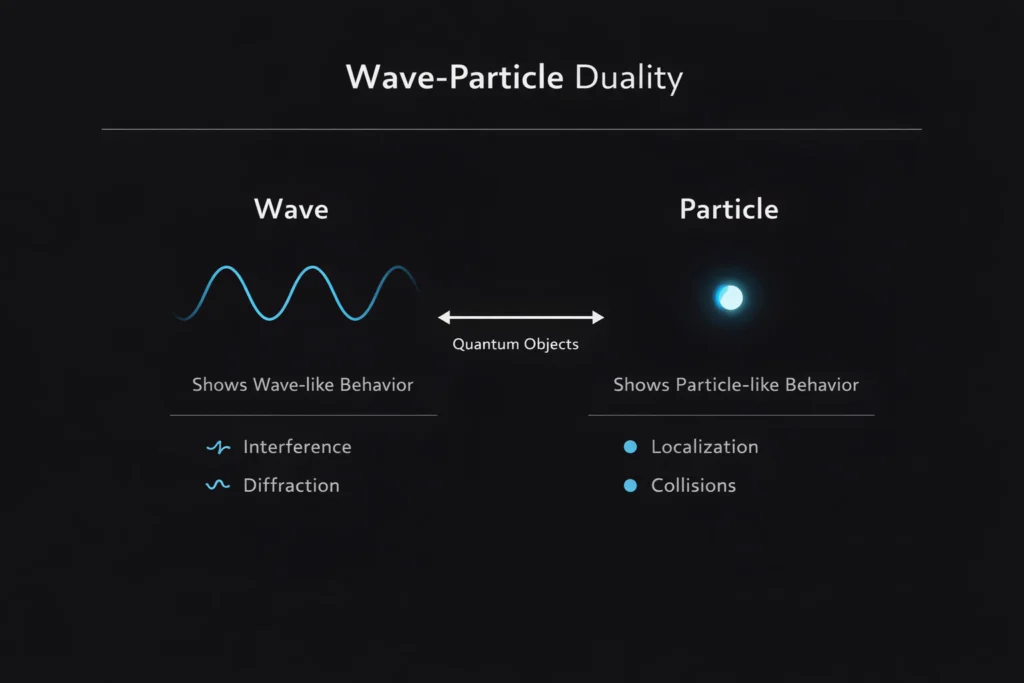
Wave-Particle Duality: Why Quantum Objects Are Both at Once
The Question That Divided Physics for Two Centuries
Is light a wave or a particle? This question occupied some of the greatest scientific minds in history for over two hundred years, and the arguments on both sides were serious, well-reasoned, and backed by solid experimental evidence. Isaac Newton believed light was made of tiny particles which he called corpuscles. Christian Huygens believed light was a wave. For much of the 17th and 18th centuries the debate went back and forth with neither side able to deliver a knockout blow.
Then in the early 19th century the wave camp seemed to win decisively. Thomas Young’s double slit experiment in 1801 produced interference patterns that only waves could explain. Augustin-Jean Fresnel developed a comprehensive mathematical wave theory of light that predicted diffraction and interference with remarkable precision. James Clerk Maxwell showed in 1865 that light is an electromagnetic wave, an oscillating combination of electric and magnetic fields propagating through space at the speed c. By the end of the 19th century the matter seemed settled beyond any reasonable doubt. Light is a wave. The particle theory was dead.
Then Einstein killed the wave theory. Or rather, he showed that the wave theory by itself was incomplete. His 1905 explanation of the photoelectric effect demonstrated that light sometimes behaves as though it is made of discrete particles, each carrying a specific fixed amount of energy determined by its frequency. A single photon interacts with a single electron in a localized, instantaneous, particle-like way. No wave picture could account for this.
So which is it? The answer that quantum mechanics eventually arrived at is both. And that answer, strange as it is, turns out to be not just a compromise or a cop-out but a profound truth about the nature of reality at the quantum scale. Wave-particle duality is not a problem to be solved or a paradox to be resolved. It is a fundamental feature of how the universe works.
Light as a Wave: The Evidence
The evidence for the wave nature of light is overwhelming and comes from many independent directions.
Young’s double slit experiment is the classic demonstration. Shine a light source at a barrier with two narrow slits cut in it and look at the pattern that appears on a screen placed behind the barrier. If light were made of particles, you would expect to see two bright patches on the screen directly behind the two slits, just as you would see two patches of sand if you threw handfuls of sand through two holes in a fence.
But that is not what you see. Instead you see an interference pattern, a series of alternating bright and dark bands spread across the screen. The bright bands appear where waves from the two slits arrive in phase with each other, meaning their peaks and troughs line up and they reinforce each other. The dark bands appear where waves from the two slits arrive out of phase, meaning a peak from one slit coincides with a trough from the other and they cancel each other out. This is interference, and interference is a phenomenon that only waves can produce.
Diffraction is another wave phenomenon that light clearly exhibits. When light passes through a small opening or around an obstacle, it bends and spreads out in ways that depend on its wavelength. This is impossible if light is simply a stream of particles traveling in straight lines. It requires that light has a wavelength and behaves as a wave at the opening.
The photoelectric effect itself does not disprove the wave nature of light. It just shows that the wave picture alone is insufficient. Light still produces interference patterns. Light still diffracts. Light still has a wavelength and a frequency. These are wave properties and they are real.
Light as a Particle: The Evidence
The evidence for the particle nature of light is equally solid and equally undeniable.
The photoelectric effect, as discussed in the previous article, shows that light delivers energy in discrete indivisible packets. A single photon transfers all its energy to a single electron in one instantaneous interaction. The energy of that photon depends on frequency, not on intensity. This is completely inconsistent with the continuous, spread-out energy delivery of a classical wave.
The Compton effect, discovered by Arthur Compton in 1923, provided further evidence. When X-rays scatter off electrons, the scattered X-rays have a longer wavelength than the incident X-rays. The amount by which the wavelength increases depends on the angle of scattering in a way that can be explained perfectly if you treat the X-ray photon as a particle with momentum colliding with an electron according to the laws of conservation of momentum and energy. The wave theory of light cannot explain this wavelength shift at all. The particle picture explains it exactly.
When extremely dim light hits a detector, the signal does not come in as a smooth continuous flow. It arrives as a series of individual discrete clicks, each one corresponding to a single photon being detected. Each photon arrives at a specific location at a specific time. This is exactly how particles behave, not how waves behave.
The Double Slit Experiment with Single Photons
The truly mind-bending aspect of wave-particle duality becomes clear when you repeat Young’s double slit experiment but dim the light source so much that only one photon passes through the apparatus at a time. You might send one photon per minute, or one per hour. Each photon is detected as a single point on the screen, a tiny localized impact, exactly like a particle.
After many photons have been detected individually over a long period of time, you look at where all those individual point detections have accumulated. And you find an interference pattern. The same alternating bright and dark bands that appear when you shine a bright continuous beam through the slits. Somehow each individual photon, all by itself, with no other photons present to interfere with, produces an interference pattern when many of them are accumulated.
The only way to make sense of this is to say that each individual photon passes through both slits simultaneously, interfering with itself, and the interference determines the probability of where that photon will be detected on the screen. Where the interference is constructive, there is high probability of detection and bright bands accumulate. Where the interference is destructive, there is low probability of detection and dark bands accumulate.
This is not a matter of our ignorance about which path the photon took. It is not that the photon really went through one slit and we just do not know which one. If you set up detectors at the slits to find out which one the photon goes through, the interference pattern disappears immediately and you are left with two bright patches behind the two slits. The act of measuring which path the photon takes destroys the interference. The photon behaves as a particle when you look at which path it takes, and as a wave when you do not. The behavior depends on what you choose to measure.
de Broglie’s Hypothesis: Matter Has Wavelengths
In 1924 a French physics graduate student named Louis de Broglie made a proposal in his doctoral thesis that seemed almost absurdly bold. If light, which everyone had thought was a wave, could behave as a particle, then perhaps particles, which everyone had thought were particles, could behave as waves.
De Broglie proposed that every moving particle has an associated wavelength, which is now called the de Broglie wavelength:
lambda = h / p
Where lambda is the de Broglie wavelength, h is Planck’s constant (6.626 x 10^-34 joule seconds), and p is the momentum of the particle (mass times velocity for a non-relativistic particle). The more momentum a particle has, the shorter its de Broglie wavelength.
This was an extraordinary proposal. Electrons, protons, atoms, and even large molecules have wavelengths? The idea seemed almost mystical. But de Broglie was not just speculating wildly. He reasoned by analogy with light and pointed out that just as Einstein had shown that wave-like light has particle-like properties, it was natural to ask whether particle-like matter might have wave-like properties.
The PhD committee at the University of Paris was not sure what to make of the thesis. They consulted Einstein, who said the idea was interesting and showed a veil being lifted from a great mystery. They passed de Broglie and awarded him the doctorate.
Three years later in 1927, Clinton Davisson and Lester Germer at Bell Labs in the United States were conducting experiments on how electrons scatter off nickel crystal surfaces. They accidentally heated their nickel target and changed its crystal structure, and found that the electron scattering pattern changed dramatically. When they analyzed the new pattern they realized the electrons were producing a diffraction pattern, the same kind of pattern that X-rays produce when scattered off crystals. The electrons were behaving as waves with exactly the wavelength de Broglie’s equation predicted for their momentum.
Independently and simultaneously, George Paget Thomson in Britain was performing similar experiments by shooting electrons through thin metal foils and observing the diffraction rings they produced on photographic film. He confirmed that electrons have wave-like properties with exactly the predicted wavelength.
De Broglie was awarded the Nobel Prize in Physics in 1929 for his hypothesis. Davisson and Thomson shared the Nobel Prize in 1937 for experimentally confirming it. Incidentally Thomson’s father J.J. Thomson had won the Nobel Prize in 1906 for discovering that the electron is a particle. Father and son each won a Nobel Prize, one for showing the electron is a particle and one for showing it is a wave.
Why We Do Not See Wave Behavior in Everyday Life
If everything has a de Broglie wavelength, why do we not see wave behavior in everyday objects? Why does a tennis ball not diffract when it goes through a doorway? Why does a car not produce an interference pattern when it drives through two adjacent lanes?
The answer is that wave effects only become significant when the wavelength is comparable to the size of the obstacles or openings the object interacts with. For everyday objects the de Broglie wavelength is incomprehensibly small.
Consider a tennis ball with a mass of 0.058 kilograms moving at 50 meters per second. Its momentum is 0.058 x 50 = 2.9 kg.m/s. Its de Broglie wavelength is h divided by p, which is 6.626 x 10^-34 divided by 2.9, approximately 2.3 x 10^-34 meters. That is about 10^19 times smaller than a proton. There is no physical object small enough to cause diffraction of a tennis ball. Wave effects are completely unmeasurable for any macroscopic object.
For an electron moving at a typical speed in a cathode ray tube, about 10^7 meters per second, the de Broglie wavelength is on the order of 10^-10 meters, which is about the same as the spacing between atoms in a crystal. This is why electrons diffract off crystal surfaces and why electron microscopes can image individual atoms. The wavelength of the electrons in an electron microscope is much smaller than the wavelength of visible light, allowing much finer detail to be resolved.
For neutrons, protons, and small atoms the de Broglie wavelength at thermal energies is also on the atomic scale, which is why neutron diffraction is a powerful tool for studying crystal structures. For large molecules the wavelength gets very small but is still theoretically nonzero. Experiments have demonstrated interference with molecules containing over 2000 atoms, which is an astonishing demonstration of the universal applicability of wave-particle duality.
Complementarity: The Principle That Makes Sense of It All
Niels Bohr, one of the founding fathers of quantum mechanics, proposed a philosophical framework for understanding wave-particle duality that he called the principle of complementarity. The idea is that wave behavior and particle behavior are complementary aspects of quantum objects. They are both real and both necessary for a complete description of nature, but they are mutually exclusive. You can never observe both aspects simultaneously in the same experiment.
When you set up an experiment to measure wave properties, such as which slit a particle goes through in the double slit experiment, you get particle-like results and the wave properties disappear. When you set up an experiment to observe wave properties, such as the interference pattern in the double slit experiment, you get wave-like results and you lose the ability to determine which path the particle took.
Complementarity is not a practical limitation that better technology might someday overcome. It is a fundamental feature of quantum reality built into the mathematics of quantum mechanics through the uncertainty principle. Precisely determining the position of a particle, as you would do by detecting which slit it goes through, necessarily disturbs its momentum and destroys the phase coherence needed to produce interference. The two types of measurement are inherently incompatible.
Bohr saw complementarity as a new kind of logic appropriate to the quantum world, one that replaces the classical either-or with a both-and that depends on context. Light is not a wave or a particle. It is a quantum object that exhibits wave properties in some experimental contexts and particle properties in others, and a complete description of light requires both aspects even though they cannot be observed simultaneously.
The Wavefunction: Describing Quantum Objects Mathematically
In quantum mechanics the wave-like aspect of particles is described mathematically by the wavefunction, usually written as the Greek letter psi. The wavefunction is a mathematical function that assigns a complex number to every point in space and time. It evolves according to the Schrodinger equation, which is the fundamental equation of motion of quantum mechanics, analogous to Newton’s second law in classical mechanics.
The physical meaning of the wavefunction was provided by Max Born in 1926. The probability of finding a particle at a particular location when you make a measurement is proportional to the square of the magnitude of the wavefunction at that location. The wavefunction is a probability amplitude, not a physical wave carrying energy through space the way a water wave or a sound wave does. Its square gives you probabilities.
This probabilistic interpretation of the wavefunction is one of the most philosophically significant aspects of quantum mechanics. In classical physics if you know the initial conditions of a system precisely enough you can in principle predict its future behavior exactly. In quantum mechanics even perfect knowledge of the wavefunction only allows you to calculate probabilities of outcomes. The universe at the quantum level is fundamentally probabilistic, not deterministic. This was deeply troubling to Einstein, who famously objected that God does not play dice. Most physicists today accept the probabilistic nature of quantum mechanics as a fundamental feature of reality rather than a sign of incomplete knowledge.
Applications of Wave-Particle Duality
Wave-particle duality is not just a philosophical curiosity. It has direct practical consequences that underpin important technologies.
The electron microscope exploits the wave nature of electrons to image objects at atomic resolution. Because electrons at typical accelerating voltages have wavelengths much shorter than visible light, electron microscopes can resolve details thousands of times finer than optical microscopes. They have revolutionized materials science, biology, and nanotechnology by allowing direct visualization of crystal structures, protein molecules, viruses, and individual atoms.
Neutron diffraction uses the wave nature of neutrons to determine the atomic structure of materials. Unlike X-rays which interact primarily with electron clouds, neutrons interact with atomic nuclei directly and are particularly sensitive to light elements like hydrogen that X-rays barely detect. Neutron diffraction is invaluable for studying the structure of biological molecules and hydrogen-containing materials.
Quantum tunneling, which is a direct consequence of the wave nature of particles, allows particles to pass through energy barriers that they classically should not be able to cross. Tunneling is essential for the operation of tunnel diodes, scanning tunneling microscopes, and flash memory storage devices. It is also responsible for nuclear fusion in the sun, which occurs at temperatures lower than classical physics would predict because quantum tunneling allows protons to fuse even when they do not have quite enough energy to overcome the electrostatic barrier between them.
Quantum computing exploits the wave-like superposition of quantum states to perform certain calculations exponentially faster than classical computers. The interference of quantum states, a wave phenomenon, is what gives quantum computers their computational advantage for specific problems.
Frequently Asked Questions
What is wave-particle duality?
Wave-particle duality is the principle that quantum objects such as photons, electrons, and other subatomic particles exhibit both wave-like and particle-like properties depending on what kind of measurement is performed. In some experimental situations they behave as waves showing interference and diffraction. In others they behave as particles arriving at definite locations at definite times. Both aspects are real and necessary for a complete description of quantum objects.
What did the double slit experiment prove?
The double slit experiment demonstrated that particles produce interference patterns, which is a wave phenomenon, even when sent through the apparatus one at a time. This means individual particles somehow interfere with themselves, which only makes sense if each particle has wave-like properties that allow it to pass through both slits simultaneously. When detectors are placed at the slits to determine which one the particle goes through, the interference pattern disappears, showing that the act of observation affects quantum behavior.
What is the de Broglie wavelength?
The de Broglie wavelength is the wavelength associated with any moving particle, given by lambda equals h divided by p where h is Planck’s constant and p is the momentum of the particle. It shows that all matter has wave-like properties. For everyday objects the wavelength is immeasurably small. For electrons and other subatomic particles it is comparable to atomic dimensions, making wave effects experimentally observable and practically important.
Why do everyday objects not show wave behavior?
The de Broglie wavelength of a macroscopic object is so extraordinarily small, many orders of magnitude smaller than any atomic nucleus, that no physical obstacle or opening could cause diffraction or interference. Wave effects only become significant when the wavelength is comparable to the size of the features the object interacts with. For electrons this condition is met at atomic scales. For a tennis ball it is not met under any conceivable physical conditions.
What is complementarity?
Complementarity is Niels Bohr’s principle that wave and particle properties of quantum objects are mutually exclusive but both necessary for a complete description. You can measure either wave properties or particle properties in a given experiment but never both simultaneously. The choice of what to measure determines which aspect of the quantum object’s nature is revealed.
Is the electron really a wave or really a particle?
Neither description is complete by itself. The electron is a quantum object that exhibits wave properties in some experimental contexts and particle properties in others. The mathematical description of the electron in quantum mechanics, the wavefunction, captures both aspects. The wavefunction evolves as a wave according to the Schrodinger equation, but when a measurement is made the electron appears at a definite location like a particle, with the probability of each location determined by the wavefunction. The electron is something new, something that classical physics had no category for, and wave-particle duality is our best way of describing its dual nature using concepts from our everyday experience.
Frequently Asked Questions
Get physics insights delivered weekly
Join students and educators receiving expert explanations, study tips, and platform updates every Thursday.
Join others. No spam.
allphysicsfundamentals
Making physics accessible, interactive, and genuinely understandable for students at every level.