The Uncertainty Principle: Why the Quantum World Is Fundamentally Unknowable
A Limit Written Into the Laws of Nature
In classical physics the universe is in principle perfectly knowable. If you could measure the exact position and velocity of every particle at one moment in time, you could calculate the entire future history of the universe with perfect precision. Every event that will ever happen is already determined by the present state of all matter and energy. The universe is a vast deterministic machine running according to fixed laws.
Quantum mechanics destroyed this vision completely, and not just in practice but in principle. Werner Heisenberg’s uncertainty principle, formulated in 1927, showed that there is a fundamental limit built into the laws of nature on how precisely certain pairs of physical quantities can be simultaneously known. This limit is not imposed by technological constraints or by imperfect instruments. It is not a reflection of our ignorance of some deeper reality. It is a basic feature of the universe itself, woven into the mathematical structure of quantum mechanics at the deepest level.
The Mathematical Statement
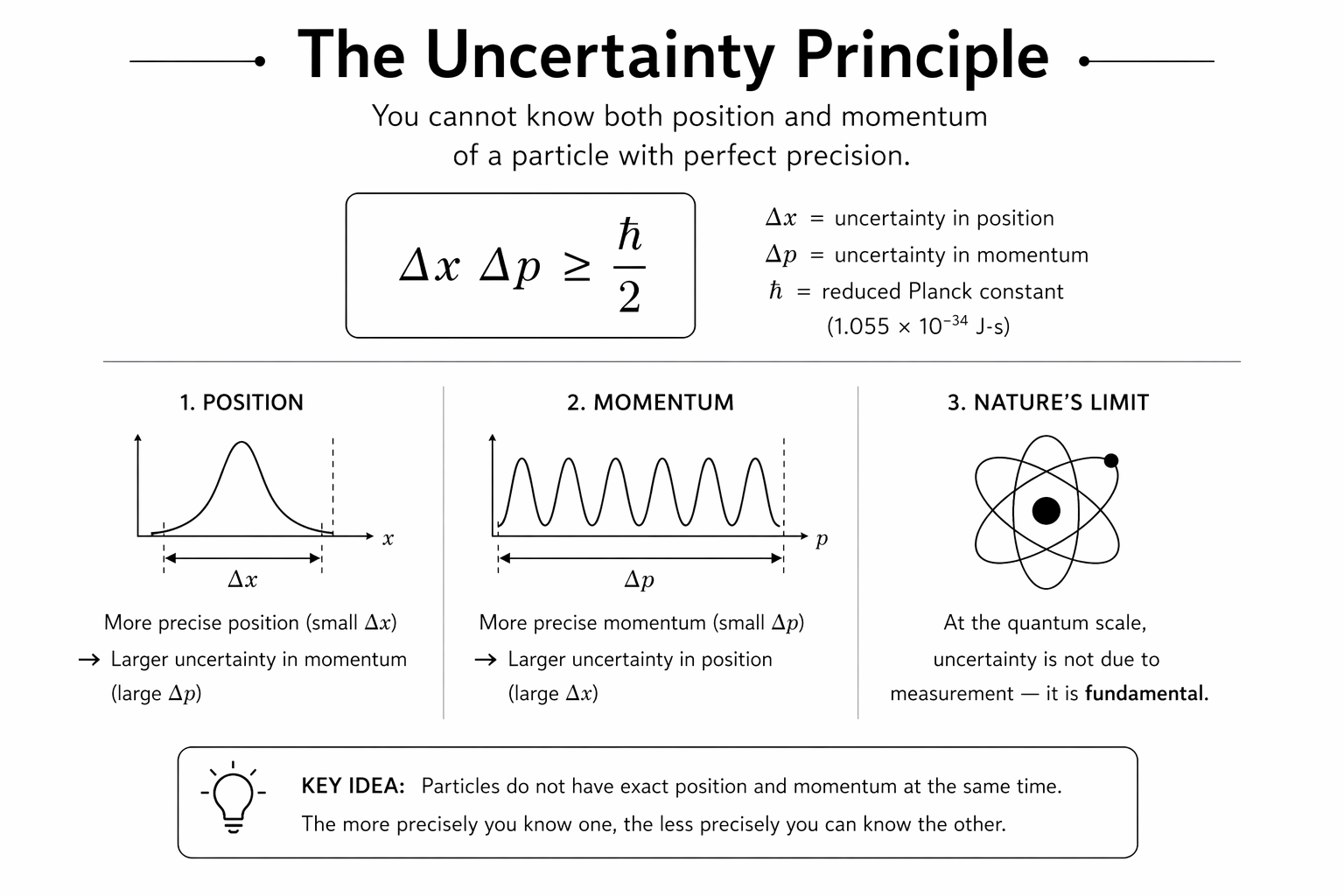
The position-momentum uncertainty principle is:
Delta x times Delta p is greater than or equal to hbar divided by 2
Where Delta x is the uncertainty in position, Delta p is the uncertainty in momentum, and hbar is the reduced Planck constant, approximately 1.055 x 10^-34 joule seconds.
The inequality says the product of position and momentum uncertainties can never be less than hbar divided by 2. Make position very precise and momentum becomes very uncertain. Make momentum very precise and position becomes very uncertain. The two are locked together by an inescapable mathematical relationship that no improvement in technology can overcome.
Delta x and Delta p are not measurement errors caused by imperfect instruments. They are the standard deviations of results you would get making many identical measurements on many identically prepared quantum systems. Even with a perfect measuring apparatus, the spread of results would still satisfy the uncertainty relation. The uncertainty is intrinsic to the quantum state itself.
It Is Not About Measurement Disturbance
The most common misconception about the uncertainty principle is that it arises because measuring a particle disturbs it. You shine light on an electron to see where it is, the photon kicks the electron and changes its momentum, and that is where the uncertainty comes from. This gives the right order of magnitude but misses the deeper point.
The real reason lies in the wave nature of quantum particles. A quantum particle is described by a wavefunction, and wavefunctions are waves. A wave localized in a small region of space must be composed of a broad range of wavelengths. A wave with a single well-defined wavelength must extend over all of space. Since momentum is related to wavelength by de Broglie’s relation, a well-localized wavefunction has poorly defined momentum and a wavefunction with well-defined momentum is spread over all space. This is a mathematical property of waves, completely independent of any measurement or disturbance.
Modern experiments have confirmed this. It is possible to prepare quantum states and make measurements that disturb the particle much less than the intrinsic quantum uncertainty. The uncertainty relation still holds, confirming it is intrinsic and not just a measurement artifact.
The Energy-Time Uncertainty Principle
There is a second form of the uncertainty principle involving energy and time:
Delta E times Delta t is greater than or equal to hbar divided by 2
A quantum state that exists for only a short time cannot have a well-defined energy. A state with very well-defined energy must persist for a long time.
This has important consequences. Excited atoms that decay quickly emit photons with a spread of energies, giving spectral lines a natural width that no instrument can eliminate. Short-lived atomic states produce broad spectral lines while long-lived metastable states produce very narrow ones.
The energy-time uncertainty principle also permits virtual particles. For a brief time Delta t, energy can fluctuate by Delta E as long as their product does not violate the uncertainty relation. Particle-antiparticle pairs can spontaneously appear from the vacuum, exist briefly, and then annihilate. The Casimir effect, a measurable attractive force between two uncharged metal plates placed close together in a vacuum, is a direct consequence of these vacuum fluctuations and has been experimentally confirmed with high precision.
Why Atoms Are Stable
The most important physical consequence of the uncertainty principle is explaining why atoms do not collapse. Classical electromagnetism predicts that an electron orbiting a nucleus should radiate energy and spiral inward, reaching the nucleus in about 10^-11 seconds. Atoms should not exist. The uncertainty principle prevents this.
If an electron were confined to a very small region near the nucleus of size Delta x, its momentum uncertainty must be at least hbar divided by (2 times Delta x). The kinetic energy associated with this momentum uncertainty increases as one over Delta x squared as the electron is confined more tightly. The attractive potential energy between electron and nucleus only decreases as one over Delta x. The kinetic energy increases faster than the potential energy decreases.
There is therefore an optimal size, a balance point where the total energy is minimized. This optimal size corresponds to the Bohr radius, approximately 5.3 x 10^-11 meters, and gives the ground state energy of hydrogen as minus 13.6 eV. The uncertainty principle stabilizes the atom by making it energetically costly to confine the electron too closely to the nucleus. This is why matter is stable. The uncertainty principle is not just a philosophical curiosity. It is the reason we exist.
Zero-Point Energy
A direct consequence of the uncertainty principle is that no quantum system can ever be completely at rest. If a particle were perfectly stationary, its momentum would be exactly zero with no uncertainty. But then its position would be completely uncertain, spread over all space. A particle confined to a definite location cannot have exactly zero momentum.
Every quantum system therefore has a minimum irreducible energy even at absolute zero temperature, called zero-point energy. For a quantum harmonic oscillator the ground state energy is one half times hbar times omega, not zero.
Liquid helium is the most striking macroscopic demonstration of zero-point energy. Helium atoms are so light and interact so weakly that their zero-point motion is large enough to prevent solidification at atmospheric pressure even at absolute zero temperature. Helium is the only element that does not solidify at atmospheric pressure no matter how cold it gets. This is a direct macroscopic consequence of the uncertainty principle.
Zero-point energy also contributes to the Casimir effect, to van der Waals forces between molecules, to the stability of chemical bonds, and to numerous other physical phenomena. It is universal across all quantum systems and has no equivalent in classical physics.
Squeezed States
The uncertainty principle sets a lower bound on the product of two uncertainties but says nothing about how that uncertainty is distributed. It is possible to make one uncertainty very small at the expense of making the other very large, as long as their product stays above the minimum. States with reduced uncertainty in one observable are called squeezed states.
Squeezed states of light have important practical applications. Gravitational wave detectors like LIGO are sensitive enough to measure displacements smaller than a proton across a four kilometer arm. Their sensitivity is ultimately limited by quantum noise in the laser light. By using squeezed light states with reduced quantum uncertainty in the relevant observable, LIGO and the Virgo detector can push their sensitivity below the standard quantum limit. The latest versions of these detectors now routinely use squeezed light in operation, representing one of the most sophisticated real-world applications of the uncertainty principle in modern experimental physics.
Philosophical Implications
The uncertainty principle raised profound questions about the nature of reality that are still debated today. If a particle does not have a definite position and momentum simultaneously, what does this mean about what is real before a measurement is made?
The Copenhagen interpretation, developed by Bohr and Heisenberg, holds that quantum mechanics is complete and that asking about unmeasured quantities is meaningless. A particle does not have a definite position before measurement. Physical reality consists only of measurement outcomes.
The many worlds interpretation proposes that all possible outcomes actually occur, each in a different branch of a vast quantum multiverse. There is no wavefunction collapse. The uncertainty principle reflects the spreading of the wavefunction across branches.
Hidden variable theories attempt to restore classical determinism by postulating that particles do have definite positions and momenta guided by a hidden field. Bell’s theorem and subsequent experiments have ruled out local hidden variable theories, severely constraining this approach.
All interpretations agree on one thing. The uncertainty principle is correct, its predictions are confirmed by experiment with extraordinary precision, and it represents a fundamental and permanent departure from the classical picture of a perfectly knowable deterministic universe.
Frequently Asked Questions
What is the Heisenberg uncertainty principle?
A fundamental law of quantum mechanics stating that position and momentum cannot both be known precisely at the same time. The product of their uncertainties is always greater than or equal to hbar divided by 2. This is not a limitation of technology but a fundamental feature of quantum reality built into the nature of wavefunctions.
Is the uncertainty principle just about measurement disturbance?
No. The uncertainty is intrinsic to the quantum state itself, not just a consequence of measurement disturbing the system. It arises from the mathematical properties of wavefunctions as waves, which cannot simultaneously have well-defined position and well-defined wavelength and therefore well-defined momentum.
What is zero-point energy?
The minimum energy a quantum system possesses even at absolute zero temperature. It exists because the uncertainty principle prevents a particle from being simultaneously at rest and at a definite location. It is physically real and responsible for helium remaining liquid at absolute zero and for contributions to the Casimir effect.
How does the uncertainty principle make atoms stable?
Confining an electron close to a nucleus gives it large momentum uncertainty and therefore large kinetic energy. This kinetic energy cost of confinement balances the electrostatic attraction at the Bohr radius, preventing collapse. The uncertainty principle is literally why matter is stable.
What is the energy-time uncertainty principle?
Delta E times Delta t is greater than or equal to hbar divided by 2. A state that exists briefly cannot have well-defined energy. This explains the natural width of spectral lines, permits virtual particles and vacuum fluctuations, contributes to the Casimir effect, and underlies Hawking’s prediction that black holes slowly evaporate by emitting radiation.
What are squeezed states?
Quantum states with reduced uncertainty in one observable at the expense of increased uncertainty in the conjugate observable. They do not violate the uncertainty principle but redistribute uncertainty. Squeezed light is now used in gravitational wave detectors like LIGO to push measurement sensitivity below the standard quantum limit.
Frequently Asked Questions
Get physics insights delivered weekly
Join students and educators receiving expert explanations, study tips, and platform updates every Thursday.
Join others. No spam.
allphysicsfundamentals
Making physics accessible, interactive, and genuinely understandable for students at every level.