Quantum Mechanics Basics: The Physics of the Very Small

Why Classical Physics Breaks Down at the Atomic Scale
For most of human history, the universe seemed to operate according to clear and predictable rules. You push something and it moves. You know where it is and how fast it is going. Given enough information about the present state of a system you can calculate its future behavior with perfect precision. This is the world of classical physics, the physics of Newton and Maxwell, and it works extraordinarily well for everything we can see and touch in everyday life.
But at the beginning of the 20th century physicists started probing deeper into matter, into the world of atoms and the particles that make them up, and they found something completely unexpected. The rules that governed the large-scale world simply did not apply at the atomic scale. Electrons in atoms did not behave like tiny planets orbiting a nucleus the way classical physics predicted they should. Hot objects did not glow with the spectrum of colors that classical theory calculated. Atoms were stable when classical electromagnetism said they should collapse in a fraction of a second. Something was deeply wrong with applying classical physics to the microscopic world.
The response to this crisis was the development of quantum mechanics in the 1920s, one of the most remarkable intellectual achievements in the history of science. In the space of just a few years, a group of brilliant physicists including Werner Heisenberg, Erwin Schrodinger, Niels Bohr, Max Born, Paul Dirac, Wolfgang Pauli, and others dismantled the classical picture of the atomic world and replaced it with something utterly new and deeply strange. Quantum mechanics is the framework that emerged from that revolution, and it has proven to be the most accurate and most precisely tested theory in the history of physics.
Quantum mechanics describes a world that operates on principles completely foreign to everyday intuition. Particles do not have definite positions or velocities until they are measured. Energy comes in discrete packets rather than flowing continuously. A system can exist in multiple states simultaneously until an observation forces it into one definite state. The outcome of a measurement can only be predicted probabilistically, never with certainty. These are not approximations or simplifications. They are fundamental features of how the universe actually works at the smallest scales.
The Crisis That Triggered the Revolution
To understand why quantum mechanics was needed, it helps to understand the specific problems that classical physics could not solve.
The first problem was blackbody radiation. A blackbody is an idealized object that absorbs all radiation that falls on it and emits radiation purely as a function of its temperature. When you heat a piece of metal it glows, first red, then orange, then yellow, then white as it gets hotter. The spectrum of light emitted, meaning how much light of each color or frequency is produced, follows a characteristic curve that depends only on temperature. Classical physics predicted this curve too, using a result called the Rayleigh-Jeans law, and the prediction worked reasonably well at low frequencies. But at high frequencies the classical prediction went catastrophically wrong, predicting that the amount of radiation emitted should increase without limit as frequency increases, reaching infinite values in the ultraviolet range. This obviously does not happen. The failure was so severe it was called the ultraviolet catastrophe.
In 1900 Max Planck found a mathematical formula that perfectly matched the observed blackbody spectrum. But to derive his formula he had to make an assumption that he himself found deeply troubling. He had to assume that the energy emitted or absorbed by the blackbody was not continuous but came in discrete chunks, integer multiples of a basic unit of energy that depended on frequency. The energy of each chunk was E = hf, where h was a new constant now called Planck’s constant and f was the frequency of the radiation. Planck thought this quantization of energy was just a mathematical trick, a computational convenience with no deep physical meaning. He was wrong. It was one of the most profound physical discoveries ever made.
The second problem was the stability of atoms. Ernest Rutherford’s experiments in 1911 showed that atoms consist of a tiny positively charged nucleus surrounded by electrons. But classical electromagnetism says that an accelerating charge radiates energy. An electron orbiting a nucleus is constantly accelerating toward the center, so it should continuously radiate electromagnetic energy, lose energy, spiral inward, and crash into the nucleus in about 10^-11 seconds. Atoms should not exist for more than a tiny fraction of a second. But obviously they do exist. Matter is stable. Classical physics had no explanation for this.
The third problem was atomic spectra. When you heat a gas of atoms and pass the light they emit through a prism, you do not see a continuous rainbow of colors. You see a series of sharp bright lines at specific frequencies, different for each element. Hydrogen always produces lines at exactly the same specific frequencies. Sodium always produces its characteristic yellow double line. Each element has its own unique spectral fingerprint. Classical physics could not explain why atoms emit light at only specific discrete frequencies rather than a continuous spectrum.
These three problems, the ultraviolet catastrophe, atomic stability, and discrete atomic spectra, were not minor puzzles around the edges of physics. They were fundamental failures of the classical framework when applied to atoms. Something completely new was needed.
Planck’s Quantum and Einstein’s Photons
The quantum revolution began with Planck’s 1900 solution to the blackbody problem. His key assumption, that energy is emitted and absorbed in discrete quanta of size hf, was the first crack in the classical edifice. But Planck himself retreated from the full implications of his discovery, hoping that quantization was just a property of how matter emits and absorbs radiation rather than a fundamental property of electromagnetic radiation itself.
Einstein went further in 1905. His explanation of the photoelectric effect required not just that energy is absorbed in quanta but that light itself consists of discrete quanta, each carrying energy E = hf. These are what we now call photons. Einstein was the first to take seriously the idea that electromagnetic radiation is quantized in its very nature, not just in its interaction with matter.
This put Planck’s constant h at the center of the new physics. Planck’s constant has the value 6.626 x 10^-34 joule seconds. Its extremely small magnitude is why quantum effects are not apparent in everyday life. The energy of a visible light photon is about 2 to 3 electron volts, which is 3 to 5 x 10^-19 joules. This is so tiny that the discrete nature of light energy is completely undetectable in ordinary experience. Only at the atomic scale where we are dealing with individual atoms and electrons does the quantization of energy become significant.
Bohr’s Model of the Atom
In 1913 Danish physicist Niels Bohr proposed a model of the hydrogen atom that used quantum ideas to solve the problems of atomic stability and discrete spectra. His model combined Rutherford’s nuclear atom with Planck’s quantum hypothesis in a bold and somewhat ad hoc way.
Bohr proposed that electrons in atoms do not orbit the nucleus in any arbitrary orbit as classical physics would allow. Instead they are restricted to specific allowed orbits, each corresponding to a specific energy level. In these allowed orbits the electron does not radiate energy despite being in circular motion, in direct contradiction to classical electromagnetism. Bohr simply postulated this as a rule without deeper justification. It worked.
When an electron jumps from a higher energy orbit to a lower energy orbit, it emits a photon whose energy equals the difference between the two energy levels:
E photon = E higher minus E lower = hf
This means light is emitted at only specific frequencies corresponding to the differences between specific allowed energy levels, which explains why atomic spectra consist of sharp lines rather than a continuous distribution.
When an electron absorbs a photon whose energy exactly matches the gap between two energy levels, it jumps up to the higher level. This explains absorption spectra, the dark lines seen when white light passes through a cool gas. The gas absorbs photons at exactly the frequencies corresponding to its allowed energy level transitions.
For hydrogen, Bohr’s model gave energy levels described by:
E n = minus 13.6 eV / n2
Where n is a positive integer called the principal quantum number (n = 1, 2, 3, …) and 13.6 eV is the ionization energy of hydrogen, the energy needed to remove the electron completely. The ground state with n = 1 has energy minus 13.6 eV. The first excited state with n = 2 has energy minus 3.4 eV. The difference is 10.2 eV, corresponding to a photon in the ultraviolet range.
Bohr’s model was a huge step forward. It explained hydrogen’s spectrum with remarkable accuracy and introduced the concept of quantized energy levels that remains central to quantum mechanics. But it had serious limitations. It only worked well for hydrogen and other one-electron systems. It could not explain the relative intensities of spectral lines, the fine structure of spectra, or why some transitions were allowed and others were forbidden. And it was internally inconsistent, mixing classical mechanics with quantum postulates in an unsatisfying way. It was a stepping stone, not the final destination.
The Wavefunction and the Schrodinger Equation
The modern mathematical framework of quantum mechanics was established in 1926 by Erwin Schrodinger and Werner Heisenberg working independently and using very different mathematical approaches. Their theories looked completely different but were soon shown to be mathematically equivalent.
Schrodinger’s approach, called wave mechanics, describes the state of a quantum system using a mathematical function called the wavefunction, typically written as the Greek letter psi. The wavefunction is a function of position and time that contains all the information about the quantum state of a particle or system. It is generally a complex-valued function, meaning its values involve the square root of negative one, which is one reason why the wavefunction itself is not directly observable.
The wavefunction evolves in time according to the Schrodinger equation, which is the fundamental equation of motion of quantum mechanics:
i hbar d(psi)/dt = H psi
Where i is the square root of minus one, hbar is the reduced Planck constant (h divided by 2 pi), d(psi)/dt is the time derivative of the wavefunction, and H is the Hamiltonian operator which represents the total energy of the system. For a particle in a potential the Schrodinger equation becomes a partial differential equation whose solutions are the allowed wavefunctions for that potential.
The physical interpretation of the wavefunction was provided by Max Born in 1926. The probability of finding a particle at a particular location x when a position measurement is made is proportional to the absolute square of the wavefunction at that location, written as |psi(x)|2. This is the Born rule, one of the fundamental postulates of quantum mechanics.
This probabilistic interpretation has profound implications. In classical physics if you know all the initial conditions you can in principle predict exactly where a particle will be at any future time. In quantum mechanics even if you know the wavefunction exactly, which represents complete knowledge of the quantum state, you can only calculate the probability of finding the particle at each location. The outcome of a measurement is genuinely random, not just unknown. The universe at the quantum level is fundamentally probabilistic.
This was deeply troubling to many physicists, Einstein included. Einstein spent decades trying to show that quantum mechanics was incomplete, that the probabilities reflected our ignorance of some deeper underlying reality with definite values. The debate culminated in the famous EPR paradox paper of 1935, in which Einstein, Podolsky, and Rosen argued that quantum mechanics must be incomplete. Subsequent theoretical work by John Bell and experiments in the 1980s and beyond have shown that Einstein was wrong and quantum mechanics is right. There are no hidden variables that would restore classical determinism. The probabilities of quantum mechanics reflect genuine randomness in nature, not ignorance.
Quantized Energy Levels
One of the most important consequences of the Schrodinger equation is that it naturally produces quantized energy levels when applied to a particle in a confined space or a potential well. This explains Bohr’s postulate about allowed orbits in a much more satisfying way.
Consider a particle confined to a one-dimensional box of length L, a simple model that captures the essential physics of quantum confinement. The walls of the box represent infinite potential barriers that the particle cannot penetrate. Solving the Schrodinger equation for this system gives wavefunctions that must fit exactly within the box, meaning they must have nodes at both walls. This requirement, analogous to the condition for standing waves on a string of fixed length, restricts the allowed wavelengths to:
lambda n = 2L / n
Where n is a positive integer (n = 1, 2, 3, …). Using de Broglie’s relation p = h / lambda, and the fact that kinetic energy equals p2 / (2m), the allowed energies are:
E n = n2 h2 / (8mL2)
These are the quantized energy levels of the particle in the box. The particle can only have these specific energies and no others. The lowest energy level with n = 1 is called the ground state. Importantly the ground state energy is not zero. Even in its lowest energy state the particle has kinetic energy, a direct consequence of the uncertainty principle as we will discuss in the next article.
The particle in a box is a simplified model but it captures the essential physics of quantum confinement. Real quantum systems like electrons in atoms, electrons in quantum dots, and nucleons in nuclei all have quantized energy levels that arise from the same underlying principle of the Schrodinger equation.
Superposition: Existing in Multiple States at Once
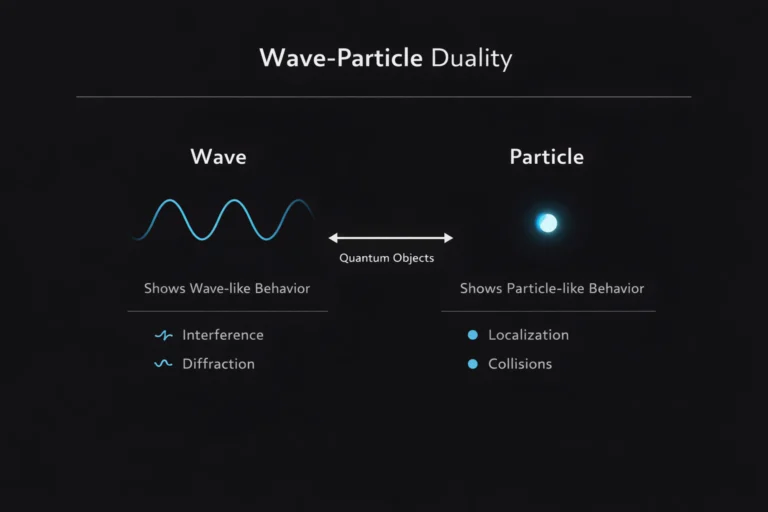
One of the most counterintuitive features of quantum mechanics is the superposition principle. A quantum system can exist in a superposition of multiple different states simultaneously, until a measurement is made that forces it into one definite state.
Mathematically this follows directly from the linearity of the Schrodinger equation. If psi1 and psi2 are both valid solutions to the Schrodinger equation for a given system, then any linear combination a times psi1 plus b times psi2 is also a valid solution, where a and b are complex numbers. A particle can be in a superposition of being at position A and position B simultaneously, or in a superposition of spin up and spin down, or in a superposition of any two or more of its allowed states.
The superposition is real and physical, not just a reflection of our ignorance. The double slit experiment demonstrates this directly. A single electron passing through the double slit apparatus is in a superposition of going through the left slit and going through the right slit simultaneously. This superposition allows the electron to interfere with itself and produce the interference pattern on the screen. If the electron were really going through just one slit or the other, with us merely ignorant of which, no interference would occur.
When a measurement is made, the superposition collapses to one of the possible outcomes with probability determined by the squared magnitudes of the superposition coefficients. If the electron is in a superposition of state 1 with amplitude a and state 2 with amplitude b, measurement gives state 1 with probability |a|2 and state 2 with probability |b|2, with |a|2 plus |b|2 equaling one.
The famous thought experiment of Schrodinger’s cat illustrates the strangeness of superposition taken to its logical extreme. A cat is placed in a box with a device that will kill the cat if a single radioactive atom decays. The radioactive atom is in a quantum superposition of decayed and not decayed. If quantum superposition applies universally, the cat should be in a superposition of alive and dead until the box is opened and an observation is made. This strikes most people as absurd, and it illustrates the measurement problem, the question of when and how quantum superpositions collapse into definite classical outcomes. This question remains one of the deepest unresolved issues in the foundations of quantum mechanics.
Quantum Tunneling
Classical physics says that a particle cannot pass through an energy barrier higher than its kinetic energy. If a ball does not have enough energy to get over a hill, it rolls back. Simple. But in quantum mechanics the situation is different because the particle is described by a wavefunction that does not abruptly stop at a barrier. Instead it decays exponentially inside the barrier and emerges on the other side with reduced but nonzero amplitude. This means there is a nonzero probability of finding the particle on the other side of the barrier even though classically it should not be able to get there. This phenomenon is called quantum tunneling.
Tunneling probability decreases exponentially with the height and width of the barrier and with the mass of the particle, which is why it is significant for electrons and protons but completely negligible for macroscopic objects. A ball does not tunnel through walls. But electrons tunnel through thin insulating barriers in tunnel diodes and flash memory devices. Protons tunnel through the electrostatic repulsion between them in the sun, making nuclear fusion possible at temperatures lower than classical physics would require. The scanning tunneling microscope uses the exponential sensitivity of tunneling current to tip-surface separation to image individual atoms on surfaces.
Alpha decay in radioactive nuclei is another example of tunneling. The alpha particle is trapped inside the nucleus by the nuclear strong force but is quantum mechanically able to tunnel through the potential barrier at the nuclear surface and escape. The rate of decay depends exponentially on the tunneling probability, which explains the enormous range of half-lives observed in different radioactive nuclei.
Spin: An Intrinsically Quantum Property
Electrons and many other subatomic particles possess an intrinsic form of angular momentum called spin that has no classical analog. Spin is not the particle physically rotating like a tiny top, despite the name. It is a purely quantum mechanical property that emerged from the Dirac equation, the relativistic generalization of the Schrodinger equation.
For electrons, spin can take only two values when measured along any axis: plus one half times hbar, called spin up, or minus one half times hbar, called spin down. This binary nature of electron spin is fundamental to the structure of matter. The Pauli exclusion principle, which states that no two electrons can occupy the same quantum state simultaneously, follows from the spin statistics theorem of quantum field theory. The exclusion principle is why electrons fill up successive energy levels in atoms rather than all collapsing to the ground state, and why the periodic table of elements has the structure it does. Without spin and the exclusion principle, matter as we know it could not exist.
Spin is also the basis of magnetic resonance imaging (MRI), one of the most important diagnostic tools in medicine. Protons in the body also have spin one half and act as tiny magnets. In a strong external magnetic field their spins precess at a characteristic frequency called the Larmor frequency. Pulsed radio frequency radiation at the Larmor frequency can flip the spins, and the energy they emit as they relax back to their equilibrium orientation can be detected and used to create detailed images of internal body structures.
The Correspondence Principle
One of the important ideas that guided the development of quantum mechanics is the correspondence principle, articulated by Niels Bohr. It states that in the limit of large quantum numbers, meaning large energies or large systems, the predictions of quantum mechanics must agree with the predictions of classical physics.
This makes sense because classical physics works extremely well for large objects. A quantum theory that gave wildly different predictions from classical mechanics for everyday objects would be clearly wrong. The correspondence principle ensures that quantum mechanics smoothly merges into classical mechanics in the appropriate limit.
For example, the quantized energy levels of a particle in a box become so closely spaced at high quantum numbers that they effectively form a continuum, just as classical mechanics predicts. The discrete spectrum of hydrogen at very high principal quantum numbers n approaches a continuous spectrum. Quantum interference effects average out over many quantum states and become undetectable for macroscopic objects.
The correspondence principle was not just a philosophical constraint. It was a practical tool that Bohr and others used to check that proposed quantum theories were heading in the right direction. Any quantum theory that violated the correspondence principle would have to be wrong.
Frequently Asked Questions
What is quantum mechanics in simple terms?
Quantum mechanics is the branch of physics that describes how matter and energy behave at the atomic and subatomic scale. It replaces the deterministic exact predictions of classical physics with probabilistic predictions based on wavefunctions. Key features include quantized energy levels, wave-particle duality, superposition, and the uncertainty principle.
What is a wavefunction?
A wavefunction is a mathematical function that completely describes the quantum state of a particle or system. The square of the wavefunction magnitude at each point in space gives the probability of finding the particle at that location when a measurement is made. The wavefunction evolves in time according to the Schrodinger equation.
What is quantum superposition?
Quantum superposition is the ability of a quantum system to exist in multiple states simultaneously before a measurement is made. When a measurement is performed, the superposition collapses to one of the possible outcomes with probability determined by the wavefunction. The double slit experiment demonstrates superposition directly through the interference pattern produced by individual particles.
What is quantum tunneling?
Quantum tunneling is the phenomenon where a quantum particle passes through an energy barrier that it classically should not be able to cross. It occurs because the quantum wavefunction does not stop abruptly at a barrier but decays through it, giving a nonzero probability of the particle appearing on the other side. Tunneling is responsible for nuclear fusion in stars, alpha radioactive decay, and is exploited in tunnel diodes and flash memory.
Why is quantum mechanics so strange?
Quantum mechanics is strange because it describes a world fundamentally different from our everyday experience. Particles do not have definite properties until measured. Outcomes are genuinely random rather than determined. Objects can be in multiple states simultaneously. None of these features appear in classical physics or everyday life because quantum effects are only significant at the atomic scale where Planck’s constant matters. Our intuitions are built from experience with macroscopic objects and simply do not apply to quantum systems.
What is the Pauli exclusion principle?
The Pauli exclusion principle states that no two identical fermions, particles with half-integer spin like electrons, can occupy the same quantum state simultaneously. For electrons in atoms this means each orbital can hold at most two electrons, one with spin up and one with spin down. This principle determines the electronic structure of atoms, the organization of the periodic table, and ultimately the stability and diversity of all matter.
Frequently Asked Questions
Get physics insights delivered weekly
Join students and educators receiving expert explanations, study tips, and platform updates every Thursday.
Join others. No spam.
allphysicsfundamentals
Making physics accessible, interactive, and genuinely understandable for students at every level.